If you follow the Stronger By Science Cinematic Universe, then you know we’ve been discussing p-ratios lately. Conceptually, p-ratios are pretty simple; when you gain weight, some of the added weight will contribute to the body’s protein reserves (that is, some will be lean mass), and the rest will predominantly be stored as fat mass. You could use a more complicated equation to calculate a “true” p-ratio value that accounts for energy densities of fat mass and fat-free mass, but for the purposes of this article, we’ll adopt a simple and practical calculation that is more commonly used in the fitness world. This approach quantifies the p-ratio as the proportion of weight that is gained (or lost) as fat-free mass, relative to the total change in body weight. In other words:
Change in fat-free mass ÷ change in total body mass = p-ratio
So, if you gain some weight and almost all of it is lean mass, you’ll have a very high p-ratio; if you gain some weight and almost all of it is fat mass, you’ll have a very low p-ratio. P-ratios can also be used for weight loss scenarios, where a high p-ratio actually becomes unfavorable for the typical lifting enthusiast who, presumably, would like to retain lean mass. Losing a bunch of lean mass but minimal fat mass would result in a high p-ratio during weight loss, whereas losing a bunch of fat mass but minimal lean mass would result in a low p-ratio. So, in general, you’d probably want to have a high p-ratio during a bulk, and a low p-ratio during a cut.
In the evidence-based corner of the fitness industry, it’s become widely accepted that high body-fat levels induce insulin resistance, which has an unfavorable impact on p-ratios. With this line of thinking, many have recommended that you should get relatively lean before you try to gain a bunch of muscle, as this will “potentiate” your lean mass gains or allow you to make relatively leaner gains during your bulking phase. However, “widely accepted” is not synonymous with “known to be true and verified by a robust body of scientific evidence.”
My interest in p-ratios began with an article back in Volume 5, Issue 1 of the MASS Research Review. A new rodent study indicated that a Western diet and a high-fat diet promoted similar degrees of obesity in mice, but only the high-fat diet significantly inhibited hypertrophy in response to muscular overload. This would indicate that gains during this rodent “bulking phase” were not negatively impacted by obesity per se, but rather by the composition of the high-fat chow, which differed from the Western diet in terms of both macronutrient profile and food sources. Of course, we never make definitive conclusions based on a single study, and we never ever make definitive conclusions based on a single rodent study due to generalizability concerns. So, this mouse study served as a springboard for a thorough review of the p-ratio concept. I’ve discussed it in MASS, on the Stronger By Science Podcast, and on the Revive Stronger YouTube channel, and now I’m putting together an article to present my “final word” (for now) on the matter. We will discuss where this whole p-ratio idea came from, how it’s commonly applied in the evidence-based fitness world, and how well the available evidence matches the hypothesis. In doing so, I’ll present brand new results from an informal participant-level meta-analysis of several studies investigating hypertrophy across a wide range of body-fat levels, and of course I’ll tie it all together with some tips for practical application. We’ve got a lot of ground to cover, so let’s dive in.
Where Did This P-Ratio Idea Come From?
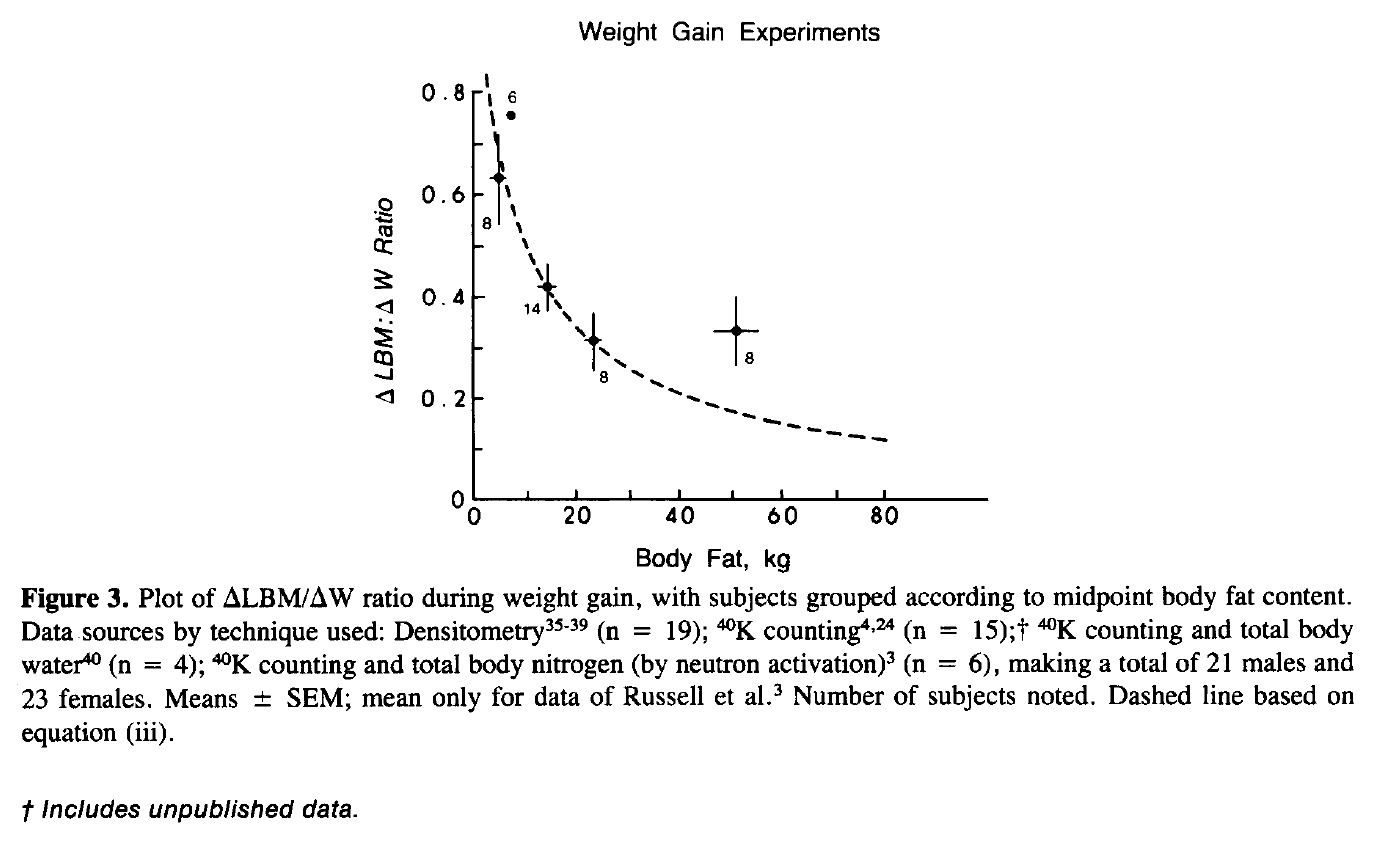
As far as I can tell, the first acknowledgement of a “p-ratio,” describing some energy-partitioning parameter pertaining to relative changes in fat mass and lean mass, dates back to a series of papers by Payne & Dugdale in 1977 (here, here, and here). But things really got exciting when the concept was expanded upon by Gilbert Forbes in 1987. Forbes argued that baseline body-fat levels were a major determinant of one’s p-ratio, such that people with higher baseline body-fat would have a very low p-ratio during overfeeding (they would gain mostly fat) and people with lower baseline body-fat would have a very high p-ratio during overfeeding (they would gain mostly lean mass). In 1987, Forbes presented the following model, which was republished in another one of his papers in 2000:
There are a few challenges when trying to apply Forbes’ model to lifters. The first hurdles simply involve interpreting the figure; it has five data points, but cites nine studies along with some amount of unpublished data. In addition, many older papers (like this one) omit study titles from their list of references, which makes it slightly more difficult to quickly track down the original studies (or at least get a sense of what they were about). To reinforce the difficulty interpreting this figure, look no further than a candid comment from Kevin Hall in his 2007 review paper:
“During the review of the present manuscript, a referee kindly pointed out that the weight gain data originally presented by Forbes in support of his theory included data from weight regain studies in anorexic patients (Forbes, 1987; Forbes, 2000). Consequently, after removing the data from the anorexic subjects with very low initial FM [fat mass], there was insufficient evidence of a relationship between the composition of weight gain and the initial FM.”
In other words, even Kevin Hall (in my opinion, an absolutely top-top-tier nutrition researcher) had a little bit of difficulty identifying exactly what types of interventions these data were derived from. But the other part of his comment brings me to the second concern: much of the data in the Forbes model is in no way generalizable to lifters.
This is not a criticism of the Forbes paper; it’s like saying that a piece of chocolate cake is the worst slice of pizza you’ve ever eaten. The Forbes model was never presented as a model for determining how to plan a bodybuilder’s bulking and cutting cycles; it was merely a cross-sectional observation about relative gains in lean mass and fat mass in people who overfed from a high or a low starting body-fat level. Notably, none of the people lifted weights, and many were overfeeding in order to recover from fairly advanced anorexia nervosa. Other data in the model came from participants who recently underwent “total starvation,” and were then refeeding from a unique physiological state in which they literally starved for up to 45 weeks, inducing fat losses of up to 66.5kg. Needless to say, this wasn’t a bodybuilding paper.
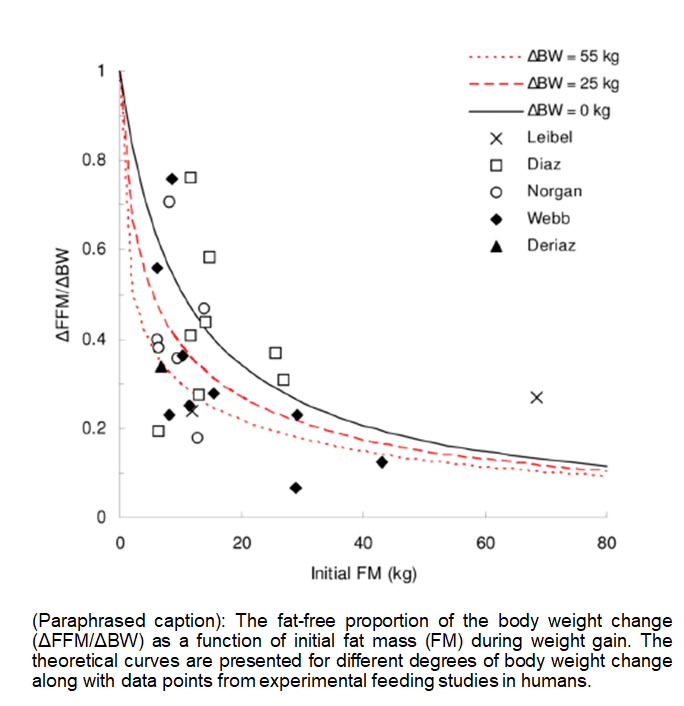
As I already alluded to, Kevin Hall expanded upon this concept in his own review paper in 2007. One of the bright spots in Kevin Hall’s approach is that he removed the anorexia data, expanded his literature search to include other studies, and found that the general premise of Forbes’ model still held up (that is, people who were leaner at baseline had higher p-ratios during overfeeding). Here is the primary figure demonstrating Hall’s findings related to overfeeding:
Props to Kevin Hall, because this figure is much, much more interpretable. However, this figure does not demonstrate the air-tight case that many evidence-based fitness folks imply. For two of the studies in the figure (Leibel et al and Deriaz et al), individual-level data were not available, so only the means were plotted. The mean baseline fat mass for Leibel et al was the highest in the figure (around 70kg), and the mean baseline fat mass for Deriaz et al was among the lowest (under 10kg). These two studies effectively span the spectrum from lowest to highest baseline body-fat levels, and the proportion of weight gained as fat-free mass was nearly equivalent (around 0.3). Looking at the individual data, imagine you’re someone who falls within the “bulk” of the data between 5-20 kg of fat mass. Based on the figure, you might gain 80% of your weight as fat-free mass, or you might gain less than 20% of your weight as fat-free mass. Most of the people applying this concept in the fitness world are wondering if they will improve their future p-ratio by losing a few kilograms before they bulk up, and starting with an initial body-fat level of 20kg or less. If we assume that these data can be generalized to plan bulking and cutting phases, it’s extremely difficult to look at these data and suggest that an individual with under 20-25 kg of fat mass could reliably expect a p-ratio benefit from virtually any magnitude of body-fat reduction.
Having said that, we still shouldn’t assume that these data are generalizable for planning bulking and cutting phases, because a few key issues persist. First of all, we are still looking at data from non-lifters. It seems implausible to expect notable lean mass gains in people with obesity who are not lifting, because there is no stimulus or physiological rationale to justify the accretion of substantial amounts of lean mass. In contrast, some leaner people may be chronically underfed, and may have some “catch-up” lean mass to gain, even in the absence of a resistance training stimulus. So, the observed relationship in the Hall models could truly be this simple in nature, and this relationship would dramatically change when you throw a resistance training stimulus into the mix. Second of all, these models are purely cross-sectional. They indicate that leaner people generally have more favorable p-ratios during weight gain (although the individual data are all over the place), but the models do not indicate that people improve their p-ratio by losing weight. As such, it’s possible that the people with obesity were more likely to develop obesity because they inherently have unfavorable p-ratios and are more susceptible to fat gain, which is very different than implying that their p-ratio became unfavorable because they developed obesity. A cross-sectional, observational study gives us no way of knowing which explanation is more credible. Third, and by far most importantly: these models work both ways.
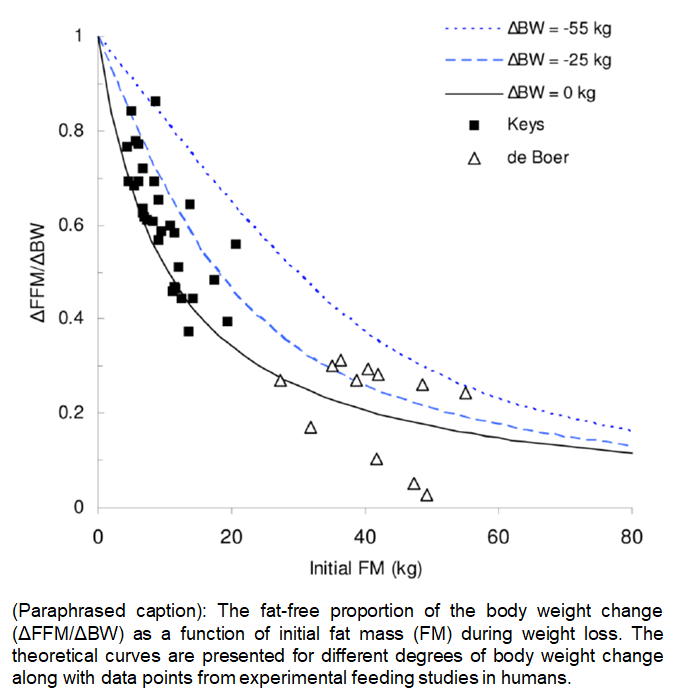
People often interpret this p-ratio relationship exclusively in the context of weight gain. They assume that they can get lean, set themselves up for a higher p-ratio in their next bulk, and enjoy a greater degree of hypertrophy and even leaner gains. However, the models by both Forbes and Hall indicate that p-ratios during weight loss are highest for the leanest people, as demonstrated in this figure from Hall’s paper:
If you are taking these models at face value, and assuming they apply to longitudinal applications in lifters (which I already disagree with), it leaves you with an unfortunate but unavoidable interpretation: if you want to lose fat to get to the sweet, sweet zone of high bulking p-ratios, you’ll have to lose lean mass to get there. If you’re trying to get to a low body-fat level for your next bulk, the models indicate that it inherently involves achieving an increasingly higher p-ratio as the preceding fat loss phase progresses (losing lean mass to a progressively greater degree as the cut progresses). In other words, you have to lose muscle to set yourself up for muscle gain. So, even if you accept the premise that these Forbes and Hall models apply to longitudinal applications in lifters, you have to arbitrarily throw out half of the model (the weight loss side) to frame it as a useful, promising strategy. Frankly, I’m not sure how you justify throwing out the unappealing side of the model and retaining the exciting side without leaning heavily on wishful thinking at the expense of thorough interpretation.
If you’re waiting for me to present the rest of the evidence in favor of the commonly asserted relationship between p-ratios and hypertrophy, you’re in for a surprise. These two review papers by Forbes and Hall, to the best of my knowledge, make up the overwhelming majority of the evidence used to promote the idea that excess body-fat will hinder your muscle-building goals, and that doing a cut or mini-cut before your next bulk will potentiate your future hypertrophy and lead to leaner gains. The idea has become so widely accepted and so rarely disputed that it can be a bit stunning to see just how flimsy the foundational evidence is, but the majority of discussions reinforcing the importance of body-fat levels for p-ratios and hypertrophy in lifters rest upon this evidence alone.
Indirect Evidence Fueling Skepticism
I’ll admit to my bias; when I first heard about this hypothesized relationship between body-fat, p-ratios, and hypertrophy, I was highly skeptical. The purported relationship didn’t seem to be compatible with the data in research areas I’m very familiar with (body composition changes in American football players and weight regain in lean people), nor was it compatible with my own anecdotal observations related to bulking, yo-yo dieting, and post-competition weight regain in physique athletes. But before we examine the indirect research related to potentially altering p-ratios, it’s helpful to logically break down what it actually means to “alter” a p-ratio. Some things in the human body truly do operate as balanced ratios; there are some carefully regulated electrochemical gradients, fluids distribute in accordance with osmotic gradients, there are balanced chemical reactions for which products and reactants reach an equilibrium, and there are reactions that proceed in ratios that match the relative availability of different enzymes. In contrast, relative gains in lean mass and fat mass during overfeeding are not truly regulated as a tightly controlled ratio. There are factors that promote or inhibit fat gain, and there are factors that promote or inhibit lean mass gain; we can calculate the observed ratio of the two after the fact, but the ratio is more of a description than a regulatory driver of physiologically coupled processes. So, if you’re altering a p-ratio during weight gain, you’re either causing fat gain, or blunting hypertrophy; the p-ratio is no more than a relative representation of these two simultaneous processes.
As a result, we can dive deeper in the p-ratio concept by evaluating the individual pieces of the p-ratio puzzle (fat gain and lean mass gain). First, we can rule out the idea that obesity causes fat gain. Spontaneous fat gain doesn’t occur; fat cells are simply the storage reservoir for extra calories that have no other metabolic fate at a given moment in time. Some have argued that extra fat mass induces insulin resistance in muscle tissue, which causes calories and nutrients to be shuttled toward fat cells instead of muscle cells, which promotes fat gain and starves the muscle cells of the nutrients and energy they need to grow. While ignoring the fact that fat cells are also susceptible to insulin resistance, this is essentially a paraphrased version of the carbohydrate-insulin hypothesis of obesity, which has been soundly rejected (and often mocked) in the evidence-based fitness world. As such, any theoretical foundation for the idea that high body-fat levels cause fat gain via insulin resistance rests on extremely shaky ground.
If something is altering a p-ratio without causing fat gain, it must intuitively be blunting hypertrophy. Anecdotally, that just doesn’t sit well with me. As Greg and I have discussed a few times on the podcast, just about everyone who is big and strong has had some phase where they implemented something approximating a “dreamer bulk” (that is, a focused bulking phase in which they dramatically over-consumed calories, stopped giving a damn about fat gain, and just packed on a ton of mass). I’ve heard many regrets about these bulking phases; some people acknowledge that they gained unnecessary amounts of fat mass, some people simply regret the unpleasant experience of being nauseatingly full and bloated for months on end, but I’ve never heard of a scenario where someone regretted a dreamer bulk because gains were hard to come by at a higher body-fat level. While some might argue that the excess fat gained during a dreamer bulk is indirect evidence of an unfavorably impacted p-ratio, I’d encourage you not to overthink it too much; more likely, the fat gain is a direct result of the obscenely large caloric surplus that defines the bulking strategy.
Assessing the Purported Mechanism
As a reminder, the primary mechanism most commonly used to support this hypothesized relationship between higher body-fat and impaired hypertrophy is that excess fat mass leads to insulin resistance in muscle cells, which shunts energy and nutrients toward fat mass and away from muscle cells, thereby favoring fat gain and attenuating muscle growth. It’s important to note that neither Forbes nor Hall incorporated any variables related to insulin or glycemic control in their p-ratio models, but this speculative mechanism took hold in the fitness world anyway. While this narrative linking excess fat mass to lower protein balance via an insulin-related mechanism may sound pretty plausible, the available evidence is far from conclusive. A 2018 review by Beals et al took a close look at several studies investigating the potential link between obesity and impaired rates of muscle protein synthesis, but examined a more comprehensive set of mechanisms involving increased intramuscular fat accumulation and chronic inflammation associated with obesity, in addition to increased insulin resistance. Their review of the literature did find that people with obesity at rest (that is, not currently doing or recovering from any exercise) had a smaller muscle protein synthesis response to dietary protein ingestion when compared to lean people at rest. However, obese people also appear to have lower rates of muscle protein breakdown, so net protein balance (the actual determinant of net accretion or loss of muscle tissue) doesn’t seem to be much different, and as we see from cross-sectional data, people who develop obesity end up with more, not less, lean mass.
This doesn’t promote a great deal of confidence in generalizing the hypothesized relationship between body-fat levels, p-ratios, and hypertrophy to active people, because the proposed mechanisms (increased insulin resistance, intramuscular fat accumulation, and chronic inflammation) should be most pronounced in rested, untrained people. While excess adiposity directly contributes to all three, they can each be offset by high activity levels and regular exercise, to some extent. There isn’t much evidence looking at these mechanisms in exercising people with varying degrees of body-fat, but the evidence that exists is mixed. I’m aware of only two such studies. The first reported that individuals with and without obesity had similar muscle protein synthetic responses to resistance exercise, while the individuals with obesity had greater insulin resistance and higher C-reactive protein levels (a marker in inflammation). So, discrepancies related to insulin resistance and inflammation were present (as expected), but these mechanisms didn’t impact the muscle protein synthesis observed in response to resistance exercise. The second reported that obese and non-obese individuals had similar increases in myofibrillar protein synthesis in response to protein ingestion alone, but non-obese people experienced a significantly higher rate of myofibrillar protein synthesis in response to the combination of resistance training and protein ingestion. These findings are difficult to interpret; obesity did not seem to unfavorably impact protein synthesis in response to protein ingestion in a non-exercised state, which appears to contradict a decent chunk of evidence in this area. Further, rates of myofibrillar protein synthesis in obese individuals were virtually identical when comparing their trained leg to their untrained leg. This suggests that resistance training had no impact whatsoever on myofibrillar protein synthesis in obese people, which seems to contradict the findings of the previously mentioned study. Taken together, there is insufficient evidence to definitively conclude that obesity impairs muscle protein balance via the proposed mechanisms (insulin resistance, intramuscular fat accumulation, and inflammation), particularly in participants who are engaged in exercise. Of course, even if this mechanistic relationship was conclusively identified in these acute scenarios, it would take another leap of faith to extrapolate that to long-term body composition changes and hypertrophy over the course of a longitudinal resistance training program.
Indirect Evidence From Athletes
In comparison to athletes, the proposed mechanisms linking body-fat to hypertrophy impairments should be more pronounced in untrained people, so impairments in muscle protein balance should theoretically be exaggerated in untrained people. However, it’s still important to check out related literature in athletes, since the people trying to apply this p-ratio idea to bulking and cutting phases are habitually engaged in exercise. The athlete literature also spans a longer time scale, looking at changes in body composition over time rather than looking at acute changes in muscle protein synthesis and breakdown, which may not correlate particularly well with longitudinal changes in muscle mass. When looking at the most muscular athletes on the planet, there is a clear pattern in which the highest fat-free mass index values are typically observed in people with relatively high body-fat levels. If excess fat mass started impairing hypertrophy as body-fat levels increased, one might think that it would be easier to continue making lean mass gains if one stayed leaner, and the most muscular athletes on the planet would tend to be relatively lean. In the sport of Sumo, there is some cool research comparing body composition among various levels of competition. If p-ratios had a strong, independent impact on lean mass accretion, one might expect that as Sumo athletes get larger and larger as they ascend to higher levels of competition, fat-free mass gains would become increasingly harder to come by. However, that’s not the case in the cross-sectional data available.
In 1999, Hattori et al published a paper documenting body composition values in Sumo athletes in four different levels of competition, listed in descending order of competitive rank: Sekitori, Makushita, Sandanme, and Shindeshi. In the Shindeshi league (mean age 18.7 years), Hattori et al reported a mean body mass of 110.7kg, with 27.4% body-fat and a fat-free mass index of 25.0 kg/m2. The Sandanme league (mean age 19.8 years) was similar: 109.3kg, 28.2% body-fat, and a fat-free mass index of 24.3 kg/m2. The highest competitive class measured in the study was the Sekitori (mean age 25.6 years); despite a much higher mean body mass (154.2kg), body-fat percentage was similar (28.6%), and the average fat-free mass index value was quite high (33.6 kg/m2). If any group of athletes were to achieve high enough body-fat levels to markedly impair their p-ratio and blunt future lean mass gains, you’d have to assume that athletes ascending to the higher ranks of Sumo would be the most likely group to demonstrate this effect. However, the data presented by Hattori et al suggest that as Sumo athletes get older and larger during their ascent to the highest levels of competition, body-fat levels remain pretty similar, and lean mass gains are plentiful. A huge limitation of this Sumo literature is that we are trying to make longitudinal inferences about cross-sectional data, so this evidence should be taken with a grain of salt. Fortunately, we can also find some evidence in longitudinal studies in collegiate American football players.
American football players represent a very convenient population for the research question at hand. The sport involves different positions with very different roles and physiological demands, and while most positions generally favor some degree of strength and muscularity, this results in a scenario where body composition is totally different from position to position. So, when you look at a college football weight room, you’ll see a bunch of people training to increase their strength and muscle mass, with some athletes having body-fat percentages as low as the single-digits and others having body-fat percentages well into the thirties. It’s also very convenient that studies almost always observe changes in different position groups within the same football team, which means the athletes are working out in the same environment, being trained by the same coaching staff, and even eating and living in the same facilities, which gives us something of a “natural experiment.” If the commonly held belief about body-fat, p-ratios, and hypertrophy holds true, we might expect that linemen (who generally have high body-fat percentages and tend to be insulin resistant) would struggle to add lean mass, or would be quite susceptible to additional fat gain in longitudinal studies. At the very least, the hypothesis suggests that linemen will need to gain disproportionately more fat mass to achieve a similar degree of lean mass gains when compared to their leaner counterparts.
Based on the research available, this doesn’t seem to be the case. Stodden et al categorized college football players into three groups: “skill” (generally lower body-fat percentages), “linemen” (generally higher body-fat percentages), and “big skill” (typically in-between). In the first year of their training after joining the team, the skill group (6.95% body-fat at baseline) gained 3.2kg of lean mass, the big skill group (11.6% body-fat at baseline) gained 3.2kg of lean mass, and the linemen group (21.1% body-fat at baseline) gained 4.6kg of lean mass. Notably, the three groups gained 3.14, 2.50, and 3.12kg of total weight over the year. So, the big skill group gained the same lean mass as the skill group while losing more fat mass in the process, and the linemen (who started with the highest body-fat percentage, by a comfortable margin) gained the most lean mass of all while simultaneously losing more fat mass than the other groups. In a similar study, Jacobson et al examined longitudinal changes while comparing only two subgroups of athletes (skill versus linemen). The linemen were 22.5% body-fat at baseline, and the skill group athletes were 8.4% body-fat at baseline. In the first year of observation, the skill players gained 6kg of fat-free mass and about 0.1kg of fat mass. In contrast, the linemen gained 6.5kg of fat-free mass and lost around 4kg of fat mass. Once again, the linemen gained more lean mass while losing more fat mass. Another study looked at body composition changes over an entire collegiate playing career in a small sample of athletes; over a four-year span, linemen gained more weight and more lean mass than non-linemen, with a greater proportion of their weight gain coming from lean mass. Of course, these are just a few studies, which may not perfectly represent the entire population of college football players. So, we shouldn’t definitely conclude that higher body-fat reliably results in better p-ratios, but these studies cast serious doubt on the idea that high body-fat levels reliably impair p-ratios.
Indirect Evidence From Weight Regain Research
Up to this point, we’ve discussed indirect research (and some anecdotes) exclusively focusing on weight gain. This line of evidence specifically addresses whether or not a person with high body-fat can successfully bulk to gain lean mass, but this overlooks a common application of the p-ratio hypothesis. A lot of people suggest that you can strategically do a cut (or mini-cut) before your next bulk in order to make it more effective than it would otherwise be. The idea is that you can longitudinally manipulate your p-ratio in order to potentiate hypertrophy after a cut; the cut would increase your insulin sensitivity, which would allow you to then achieve greater hypertrophy and gain less fat in the process (according to the hypothesis).
This is another area where the indirect evidence available just doesn’t seem to support the idea. It’s hard to imagine getting leaner or more insulin sensitive than a stage-ready physique athlete; theoretically, the observed p-ratio observed during post-competition weight regain should be as high as it gets. However, we observe quite the opposite; post-competition weight regain in natural physique athletes involves, if anything, a disproportionate amount of fat regain rather than lean mass gain. Naturally, one might argue that this evidence isn’t very generalizable because competitive physique athletes are a unique population in a unique physiological state at the time of competition. Fortunately, there is plenty of research looking at weight regain in people who aren’t shredded, and are simply trying to cut down for health-related reasons. As reviewed by Dulloo et al, we often see that fat regain is, if anything, prioritized over the regain of lean mass following weight reduction. When people are regaining weight after a successful cut, their p-ratios tend to be the same or worse than before. Unfavorable effects on an individual’s p-ratio during weight regain are more likely if that person has gotten really shredded or lost a ton of weight, but even when these unfavorable p-ratio changes are successfully avoided, we don’t reliably observe improvements in p-ratios following weight loss in the research. These longitudinal findings line up pretty well with cross-sectional studies showing that twins who have completed more intentional weight loss attempts tend to have significantly higher BMIs than their twin with fewer weight loss attempts, and showing that repeated weight loss and weight regain cycles in athletes are correlated with higher BMI values later in life. These findings also line up pretty well with real-world anecdotes; most post-competition guidelines for physique athletes aim to strategically manage the rapid fat gain that is commonly observed after competitions, and yo-yo dieting certainly isn’t embraced as an efficient means of improving body composition.
I originally wrote about this topic in MASS, and I was pretty satisfied with the case I had put together based on this indirect evidence. However, there are always going to be limitations when you’re trying to make inferences based on indirect evidence from other areas of research. For example, you could interpret the cross-sectional data differently. Higher lean mass values in Sumo wrestlers with higher body-fat percentages suggest that high body-fat doesn’t totally shut down lean mass gains, but totally shutting down lean mass gains translates to a p-ratio of zero rather than a reduced p-ratio. Since we don’t know the exact time course of fat gain and lean mass accretion, you could potentially argue that their higher body-fat percentages are a consequence of a worsening p-ratio during weight gain, and that their ability to increase lean mass per unit of fat gain may have gotten worse over time. You could also argue that American football players in different position groups have different training priorities, which could have influenced their longitudinal body composition changes. You could argue that the weight regain data is mostly from post-competition physique athletes and untrained people with high BMIs, and that neither population is a good representation of the majority of resistance-trained folks with body-fat levels closer to the middle of the spectrum. In addition, most of the studies cited so far have focused on average changes in groups rather than individual-level changes. Greg Nuckols suggested that we could improve this argument by presenting a new, original analysis of existing data, and he was right.
New Evidence: Re-Analysis of Longitudinal Resistance Training Studies
We were interested in answering a simple question: does baseline body-fat percentage predict how much fat mass or lean mass a person will gain over the course of a controlled resistance training intervention? In order to address this, we had to gather data sets from longitudinal resistance training studies reporting baseline body composition and body composition changes with individual-level data for all participants (huge thanks to Greg for tracking down and gathering these files, and huge thanks to all of the researchers for actually doing the work and making their results available). Some of the data were publicly available via journal websites, and others were directly provided to us by the researchers upon request. This wasn’t formally a “systematic review,” but we certainly weren’t cherry-picking; we just gathered all of the data sets we could find by searching, and leaned on a few colleagues who had data available, so it can be thought of as a convenience sample. In addition, we didn’t apply any kind of biased procedure for data selection; we included all of the data from all of the studies we could get our hands on.
Some studies reported various types of metrics related to muscle mass and lean tissue. For example, a study might report fat-free mass, lean soft tissue, or some estimate of skeletal muscle mass. However, as with any meta-analytic procedure, it was important to get all of the studies on “equal footing” by converting body composition data from all of these studies to a standardized metric. For all studies, body weight was provided at each time point of interest, along with either a body-fat percentage value or a fat mass value. Using this information, we could calculate fat mass and fat-free mass at each time point, which allowed us to compare all studies using the same units. Five of the studies sampled resistance-trained participants, with study durations ranging from 6-10 weeks. Two studies sampled untrained participants, but utilized longer interventions (16-20 weeks), which allows participants to spend a few weeks getting the hang of things and making some neural adaptations. We did not include any data from untrained control participants, for obvious reasons.
We are going to pool all the data together for a combined analysis, but it can be informative to first look at each study individually. For each study, we used regression analysis to determine the extent to which baseline body-fat percentage predicted changes in body composition. However, we couldn’t use the p-ratio as the outcome variable (change in fat-free mass divided by change in body weight), because this can get a little messy with real-world data. For example, if someone “recomps” (gains muscle while losing weight), the simplistic p-ratio calculation becomes negative. Similarly, if someone loses muscle while gaining weight, they’d also have a negative p-ratio. This is unfavorable, because a negative p-ratio could simultaneously represent the best and worst possible outcomes. So, we made up our own “lean gains” metric, which is simply the change in fat-free mass minus the change in fat mass. If someone gained a ton of lean mass in addition to a little bit of fat mass, their “lean gains” value would be positive. If someone gained a ton of lean mass without gaining fat, it would be higher, and it would be even higher if they managed to gain a bunch of lean mass while losing fat in the process. The result is an outcome metric that rewards gains in lean mass while numerically penalizing gains in fat mass, resulting in a number that goes up when you’re making lean gains, and fails to go up if you’re struggling to add lean mass or gaining a high degree of fat mass relative to your gains in lean mass.
For our regression analysis, this “lean gains” metric was the dependent variable, and the predictor variable was baseline body-fat percentage. Characteristics of the studies included, and the regression results for each individual study, are provided in Table 1. Below the table, I’ll provide a detailed explanation of how to interpret the findings.
| Table 1. Characteristics and results of individual studies | ||||||
| Study | N (% male) | Duration | Baseline BF% range |
Within study slope [95% CI] |
Within study p |
Within study R^2 |
| Helms et al | 21 (100%) | 8 weeks | 4.1 – 21.3 | -0.04 [-0.18, 0.09] |
0.500 | 0.024 |
| Batrakoulis et al | 28 (0%) | 20 weeks | 38.0 – 53.2 | -0.15 [-0.53, 0.22] |
0.410 | 0.026 |
| Schoenfeld et al | 21 (100%) | 10 weeks | 7.3 – 27.8 | 0.24 [0.03, 0.46] |
0.026 | 0.236 |
| Klemp et al | 16 (100%) | 8 weeks | 5.8 – 20.2 | 0.21 [-0.18, 0.61] |
0.271 | 0.086 |
| Baz-Valle et al | 18 (100%) | 8 weeks | 9.3 – 18.2 | 0.07 [-0.38, 0.51] |
0.762 | 0.006 |
| Feito et al | 26 (35%) | 16 weeks | 16.7 – 44.3 | 0.02 [-0.19, 0.23] |
0.835 | 0.002 |
| Haun et al | 31 (100%) | 6 weeks | 9.5 – 27.1 | -0.02 [-0.18, 0.15] |
0.815 | 0.002 |
It’s easiest to explain how to interpret the regression results by using an example, so let’s walk through the first study (Helms et al). The regression slope was -0.04. This means that for every one-unit increase in baseline body-fat percentage, we expect the “lean gains” metric to go down by 0.04kg. So, if a person started the study at 16% body-fat, we would expect their lean gains value to be 0.04kg lower than a person who started the study at 15% body-fat, on average. The 95% confidence interval spanned from -0.18 to 0.09. Recall that each study looks at data in a sample of participants, which was drawn from the larger population of all eligible participants; our 95% confidence interval tells us that we are 95% sure that our interval ranging from -0.18 to 0.09 covers the true slope in the population from which the data were sampled (we don’t know exactly where the true slope value is, but we’re 95% confident that our interval overlaps with it). Our p-value (not to be confused with p-ratio) for this slope is 0.500; this means that if we were to assume that the “true” slope in this population is exactly zero, there’s a 50% probability that we’d observe a slope this different (or more different) from zero. When the p-value gets below 0.05 (or 5%), we typically become skeptical of the assumption that the true slope is actually zero, and we conclude that the slope is “statistically significantly different” than zero (in other words, we would conclude that baseline body-fat percentage has a statistically significant relationship with the lean gains outcomes observed). Finally, the R2 value was 0.024. This means that 2.4% of the variance in “lean gains” can be explained by baseline body-fat percentage. If the R2 value were 0, then baseline body-fat would have no relationship whatsoever with an individual’s ability to make lean gains. If the R2 value were 1, then we could perfectly predict an individual’s “lean gains” value based on their baseline body-fat, which would reinforce the idea that the two variables are highly, highly related to each other.
Looking through the individual results, 5 of the 7 studies show a whole bunch of nothing. For those five studies, the slopes were close to zero, the p-values were high, and the R2 values indicate that baseline body-fat percentage could explain less than 3% of the variance in lean gains achieved in response to the training interventions. You could maybe argue that Klemp et al found a noteworthy relationship between baseline body-fat percentage and lean gains, with a slope of 0.21 (for example, a person with 16% body-fat at baseline would be expected to have a lean gains value 0.21kg higher than a person with 15% body-fat at baseline). However, this was not statistically significant (p = 0.271), and baseline body-fat could explain only 8.6% of the variance in lean gains. Only the results by Schoenfeld et al were statistically significant, with a slope of 0.24. In that particular study, baseline body-fat percentage explained a non-negligible 23.6% of the variance in lean gains. So, if we look at individual results, it seems that higher baseline body-fat percentage either doesn’t impact one’s ability to make lean gains, or actually improves one’s ability to make lean gains, which is consistent with a p-ratio improvement.
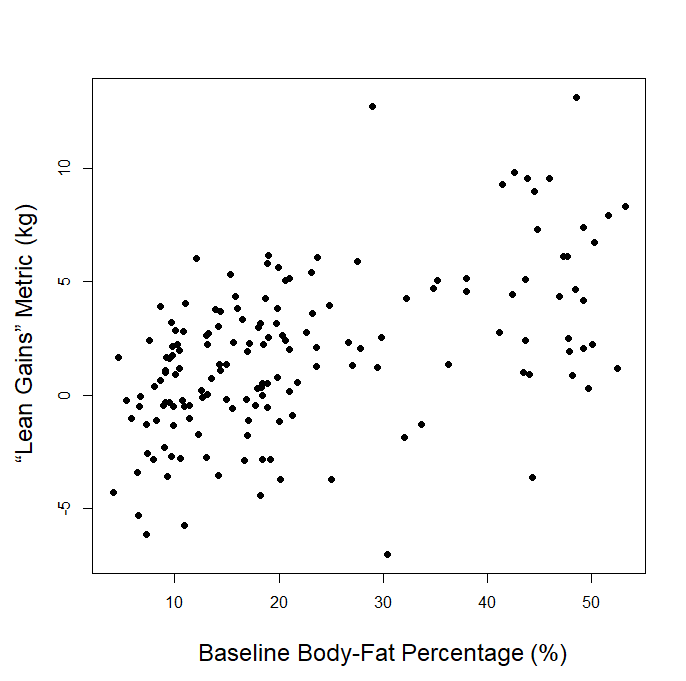
Of course, one of the main benefits of aggregating all of these data is that we can combine them for an overall summary of the literature, just as we would do with any other meta-analytic approach. If we just dump all of the data points into a single plot with baseline body-fat percentage on the x-axis and lean gains on the y-axis, this is what we get:
It appears that the slope is positive, which not only fails to support, but directly contradicts the p-ratio hypothesis commonly held in the evidence-based fitness world. If high body-fat negatively impacted p-ratios, we should see a negative slope. After visualizing the data, it might be tempting to run a standard regression model including all of the data. If we did, we’d find that the slope of the regression line is positive and significantly different from zero, with a very, very low p-value (like, more than ten zeroes after the decimal point). However, I won’t present those exact values, because they’d be misleading. If we jam these data into a standard regression model, we have to assume that 0% of the variance in lean gains is attributable to factors that differ among studies (in other words, we’d have to assume that the studies are perfectly homogeneous, such that two data points from the same study are no more similar to each other than two data points from two different studies). In reality, about 36% of the variance in lean gains is attributable to factors that differ among studies, so we ought to reject that assumption. Fortunately, we can move forward by using a linear mixed model with random intercepts, which allows participants from each of the different studies to share an intercept that varies from study to study (we could have also allowed for slopes to vary among studies, but the random intercepts model was a better fit for the data).
The results of the linear mixed model indicate that there was a positive, statistically significant slope describing the relationship between baseline body-fat percentage and lean gains (slope = 0.10, p = 0.006). So, we would expect a person with 16% body-fat at baseline to have a better lean gains outcome than a person with 15% at baseline, by about 0.1kg. Our original question was whether or not baseline body-fat influences one’s ability to make lean gains (or, in other words, influences their p-ratio). The commonly held belief is that higher body-fat impairs gains, but the model actually shows a significant effect in the opposite direction, with higher baseline body-fat predicting better lean gains.
That’s interesting, but we can dig deeper to gain a better understanding of this relationship. As I mentioned previously, we really ought to consider lean mass accretion and fat gain as two independent processes, with different stimuli and different regulatory mechanisms. When we use linear mixed models to look at gains in fat-free mass, we see that baseline body-fat has virtually no effect on fat-free mass increases in this data set (slope = 0.0045, p = 0.797). In contrast, when we use linear mixed models to look at gains in fat mass, we observe a statistically significant relationship with a negative slope (slope = -0.095, p = 0.001).
These results indicate that people with relatively low and relatively high body-fat can gain fat-free mass to a pretty similar degree in response to resistance training. However, very lean people should accept that fat mass will probably need to increase a little bit to facilitate muscle gain, and they may need to get up to a slightly more comfortable body-fat level before lean mass gains really start to accumulate (for example, every single person under 8% body-fat at baseline had some degree of fat gain, and only one of them gained more than 1kg of fat-free mass). In contrast, people with higher baseline body-fat have a greater capability to “recomp” and are more likely to simultaneously lose a little bit of fat while gaining muscle. It’s important to recognize that these results do not conflict with the laws of thermodynamics or the concept of energy balance dictating weight changes; they simply reflect the fact that the body has both short-term and long-term sensors of energy availability which can influence the regulation of anabolic processes, and that muscle hypertrophy and fat storage are influenced by some distinct regulatory mechanisms. In addition, none of these studies exerted tight control over total daily energy expenditure or daily energy intake, so subconscious alterations in diet or physical activity habits could have contributed to some of the fat loss observed in participants with higher baseline body-fat levels (but that would make it even more impressive that subjects with greater adiposity gained similar amounts of fat-free mass).
At this point, you still might not find the argument presented in this article to be sufficiently convincing. Admittedly, there are limitations to each component of the argument. As discussed previously, some of the evidence focuses on differences in muscle protein synthesis, which may not necessarily correlate with eventual differences in lean mass. Some of the evidence is cross-sectional, which may not reflect longitudinal changes. Some of the evidence compares American football players in different position groups, who may have different training priorities. We aggregated and analyzed raw data tracking longitudinal hypertrophy across body-fat levels ranging from <5% to >50%, but you may feel that this combined analysis was confounded by differing study characteristics, such as training status, training programs, biological sex, or study duration. However, my question is this: if this relationship between body-fat, p-ratios, and hypertrophy is both true and independently impactful enough to influence our training and nutrition decisions, why the hell is it so hard to find? Why does it seem to be so incompatible with so many different areas of research, and why should we abandon all of the evidence laid out here in favor of two cross-sectional review papers about overfeeding in people who aren’t lifting?
A Simpler, More Justifiable Model
When looking at the shaky theoretical footing of the concept, the totality of the indirect evidence, and the results of our new analysis, I find it very difficult to suggest that anyone needs to lose fat in order to “potentiate” hypertrophy or improve their p-ratio in their next bulking phase. I can’t say that the argument contained in this article “conclusively disproves” the idea, but that’s not how any of this is supposed to work anyway.
In science, we generally embrace the concept of parsimony, which encourages us to adopt the simplest explanation that fits the available evidence. This leads us toward generally embracing the null hypothesis, unless we are convincingly compelled to reject the null hypothesis (this is the theoretical underpinning of 99.9% of the statistical tests you’ve ever seen in a research paper). The null hypothesis is the default assumption that no relationship exists between two variables (in this case, that your starting body-fat level does not influence your ability to make gains in response to resistance training). Of course, we’re always happy to reject the null hypothesis when provided convincing evidence, but the burden of proof falls on those who wish to assert that the null hypothesis is false. There are innumerable hypotheses that cannot be irrefutably disproven due to a lack of evidence, yet lack enough evidence to substantiate them. If we follow every unsubstantiated hypothesis simply because it hasn’t been conclusively disproved, we’re going to be wrong way more often than we’re right. For this reason, we need sufficiently strong evidence to convince us that one should lose body-fat to potentiate hypertrophy and improve their p-ratio. This level of evidence has not been provided, and the hypothesis seems to be incompatible with the scientific evidence that is currently available.
In the interest of parsimony, I will provide a simpler conceptual model that more reliably fits the available evidence while requiring fewer assumptions that lack sufficient support. First, we can tackle the Forbes and Hall models, which involve no resistance training stimulus. In this scenario, accretion of lean mass is related to the stimulus for lean mass accretion, coupled with the provision of sufficient nutrients and energy intake to accommodate lean mass accretion. When we look at very, very lean individuals in these models, a stimulus for lean mass gain exists, even in the absence of resistance training. For example, patients with anorexia nervosa in the Forbes model likely induced significant atrophy of muscle tissue and other lean tissues as their medical condition progressed, so there is a biological stimulus for the reversal of this atrophy. Looking at the other end of the spectrum, individuals with obesity gained fat quite efficiently when overfed. People with obesity tended to have above-average levels of fat-free mass, so a robust stimulus for fat-free mass accretion is absent. In addition, we know that individuals prone to obesity more readily gain fat mass in controlled overfeeding studies, while obesity-resistant individuals have robust defenses against fat gain in response to the exact same overfeeding interventions. So, the efficient fat gain in obese individuals in the Forbes and Hall models might not only relate to the absence of a stimulus for lean mass accretion, but also an inherent individual-level characteristic in people with obesity that favors efficient fat gain. In other words, obesity didn’t worsen their p-ratio; they developed obesity because they naturally have a low p-ratio, and they had no reason (stimulus) to add substantial amounts of fat-free mass at an elevated body weight.
This understanding of the p-ratio as an intrinsic characteristic that varies (to some extent) from person to person is compatible with the ideas presented in the fantastic 2018 review by Dulloo et al. Having said that, an individual’s p-ratio is modifiable. If you implement an absolutely enormous calorie surplus, you’re gaining more fat than you would with a modest calorie surplus. As you progress from being super-shredded to extra-super-shredded, your likelihood of losing lean mass increases, and gains in fat-free mass are harder to come by (likely due to changes in hormone concentrations and physiological indicators of chronic energy status). Finally, as Dulloo describes in detail, an individual’s p-ratio can be either unchanged or unfavorably impacted in the context of weight regain, as fat may be preferentially regained after individuals get extremely lean or lose substantial amounts of weight. Of course, there is arguably no p-ratio modifier more impactful than resistance training, which is entirely absent from the Forbes and Hall models, and most directly relevant to (presumably) everyone reading this. So, here’s the climactic reveal of my model for how lifters should view the relationship between body-fat, p-ratios, and making gains:
Over a given time period, the amount of weight you gain or lose will be dictated by the magnitude of your caloric surplus or deficit. Your ability to gain lean mass will depend on whether or not your body has high enough energy availability to fuel muscle hypertrophy (which is an energy-intensive process), how effectively your hypertrophy-promoting resistance training program is designed, how readily you gain muscle in response to resistance training (which varies from person to person and throughout a person’s lifting career), and the degree to which you are consuming the nutrients that are required for supporting muscle hypertrophy. Your changes in fat mass will simply be the difference between these two simultaneous processes (changes in body weight and changes in lean mass). It’s simple, straightforward, and seems to fit the evidence quite well, including the unique circumstances at the upper and lower ends of the body-fat spectrum in which shredded people struggle to make super-lean gains and people with higher body-fat are more capable of recomping.
Practical Application: Should You Bulk or Cut?
I am not convinced that we can directly potentiate hypertrophy or enhance our future p-ratio by losing fat. One of the reasons I’m so interested in this topic is because I see it regrettably misapplied so frequently. It seems that a lot of people who are just getting into lifting are convinced that they must begin their lifting career by getting down to 10-15% body-fat to have any chance of being successful. This is unfortunate, because the most important thing a new lifter could do is fall in love with lifting. Fat loss might be part of the equation for some, but for others, that process simply involves getting into the gym and enjoying the muscle growth and strength increases that beginners tend to experience. I think it’s very counterproductive for new lifters to feel obligated to begin their journey with a fat loss phase that they aren’t enthusiastic about, and for them to associate their first months of lifting with hunger and lethargy rather than excitement and enjoyment. It also seems like a lot of experienced lifters fear that they’re shooting themselves in the foot by letting their body-fat percentage creep above 15% during a bulk. Unfortunately, this leads a lot of lifters to “spin their wheels” for far too long, perpetually oscillating back and forth between excessively short cutting and bulking phases, and potentially forfeiting the size and strength gains that can materialize in an extended, focused mass building phase.
Don’t get me wrong – there are plenty of good reasons for lifters to cut before a future bulk, but improving their p-ratio isn’t one of them. When someone first gets into lifting, it’s very common for their dream physique to be both leaner and more muscular than their starting physique. As a result, brand new lifters often face a dilemma which compels them to make the difficult decision between bulking first or cutting first. If they’re super excited about getting lean and bulking up from there, they should feel encouraged to do so. Of course, the predicament of wanting to be both bigger and leaner certainly isn’t unique to new lifters, and intermediate and advanced lifters commonly oscillate between fat loss phases and bulking phases. At any given time, these lifters may want to do a fat loss phase because they simply feel better, perform better, or are happier with their health-related metrics at a lower body-fat level, and they’d prefer to get there before shifting their focus toward their next bulking phase. In some cases, a lifter may wish to focus on an extended bulking phase, but might already be near the highest body-fat level they’re comfortable maintaining (whether that comfort threshold is related to their health, performance, or aesthetic preferences). Given that some degree of fat gain often accompanies dedicated muscle-gaining phases, it might not be a bad idea to do a fat loss phase, which would give them some additional leeway for a little bit of fat gain to occur during the upcoming bulking phase.
In summary, there are several good reasons why a lifter would implement fat loss phases in various stages of their lifting career. However, there is insufficient evidence to suggest that losing fat will potentiate hypertrophy or improve one’s p-ratio after a lower body-fat level is achieved. A brand new lifter should not feel obligated to begin their lifting career by cutting to a magical body-fat range in order to gain muscle, and lifters should not worry that they’ll struggle to gain muscle if they cross an arbitrary body-fat threshold. Your choice to bulk or cut depends on your goals, preferences, and what you’re most excited about doing at any given time in your lifting career.
A Reminder About “Evidence-Based” Recommendations
Before wrapping up this article, I think there’s an important, generalizable lesson to be learned from the whole p-ratio saga. This idea became very popular in the evidence-based fitness world, and went largely unchallenged. However, at its core, the argument was simply an aggressive extrapolation of a speculative mechanism that was assumed to explain a small collection of cross-sectional findings in overfed people who weren’t lifting. If you look at any of the more egregious pseudoscientific claims that run rampant in the worlds of training, nutrition, or the fringes of alternative medicine, there is almost always some purported mechanism or association with a granule of truth to it; just enough of a mechanistic basis to enable extrapolation and theorizing. When there’s a gap in the literature and we’ve got no direct (or even indirect) empirical evidence to lean on, then extrapolation and theorizing based on our mechanistic understanding of the human body is all we can really do; it’s our best attempt at patching together an incomplete understanding of phenomena that haven’t been rigorously studied. However, we could weave a virtually unlimited number of plausible and logical narratives by starting with a mechanism and extrapolating from there, with only a small portion of those narratives accurately reflecting reality.
As a result, we should lean on empirical evidence whenever possible. Even if a hypothesis or idea hasn’t been studied directly, we should attempt to check the validity of the idea by seeing if it is at least compatible with indirect evidence. So, whenever you encounter an idea, first see if it has direct empirical support. If it doesn’t, ask yourself what you should expect to observe in other areas of research if the idea is true. If you can identify a related area of research with some indirect data you can lean on, dive in and see what you find. Most importantly, try to guard against becoming overconfident in ideas just because they seem logical and plausible. It can be very tempting to embrace ideas simply because they make sense, but not all things that make sense are true, and not all true things make sense.
Summary
- In the fitness industry, we often discuss an individual’s “p-ratio” as the proportion of weight that they gain or lose as fat-free mass (i.e., if you gain 3 pounds of weight and it’s all fat-free mass, your “p-ratio” is 1; if it’s all fat mass, your “p-ratio” is 0).
- It is commonly said that your p-ratio gets worse as your body-fat percentage increases due to impaired insulin sensitivity.
- As an extension, many have suggested that it’s hard to make lean gains if your body-fat level gets too high, or that you can “potentiate” hypertrophy by doing a cut or mini-cut before your next bulking phase.
- This idea is largely based on two cross-sectional review papers assessing responses to overfeeding in people who were not lifting weights.
- Studies looking at the purported mechanism for this idea are pretty inconclusive in the context of resistance training. More importantly, this idea is not supported by cross-sectional studies in athletes, longitudinal studies in lifters, or longitudinal studies in people who are regaining weight after weight loss.
- There are all sorts of good reasons to lose fat, but there’s no compelling evidence to suggest that you’ll potentiate hypertrophy or improve your ability to make lean gains by losing fat beforehand.
- Should you bulk or cut? It depends on your goals, preferences, and what you’re most excited about doing at any given time in your lifting career.