In the evidence-based fitness community, it’s commonly believed that high body-fat levels attenuate, impair, or blunt muscle growth. As a result, many tend to assume that there is strong evidence to support this idea (if there weren’t strong and conclusive evidence for this phenomenon, how could it be so widely accepted by members of the evidence-based club?). Unfortunately, this concept not only lacks scientific support, but is actually contracted by the best available evidence.
I won’t get too deep into the background information and details, as this topic has been the subject of a previous MASS Research Review article, in addition to a three-part series of follow-up articles on Stronger By Science (one, two, three). In short, the seed of this idea was planted by Forbes back in 1987 (2). Forbes published a paper discussing his observation that “p-ratios” (the proportion of weight gained as lean mass) seemed to be higher for leaner samples, and lower for samples with higher body-fat levels during purposeful weight gain. So, the leaner samples tended to gain a larger proportion of weight as lean mass. There are some statistical considerations that should cause us to hesitate before making any sweeping conclusions from this observation. For example, the observed effect was based on a figure that included only five data points representing a total of 44 participants, and it’s honestly pretty hard to track down exactly which studies are represented in the figure. In addition, the concept relies fully on cross-sectional data. The proposed model indicates that people who are lean tend to have relatively leaner gains, and that the inverse is true for people with higher body-fat. This could theoretically be an innate characteristic that contributes to these people having lower or higher body-fat levels in the first place. More importantly, if we move beyond the statistical model, the observed relationship has absolutely nothing to do with lifters. As far as I can tell, the sample was largely made up of anorexia nervosa patients, lactating women, and people who recently underwent “prolonged total starvation” to induce dramatic weight loss, and it doesn’t look like any of the participants were undergoing resistance training. In addition, the overfeeding protocols were not standardized in any way, with variable magnitudes, durations, and macronutrient profiles.
Despite these massive shortcomings, some fitness enthusiasts ran with the concept and started suggesting that lifters should intentionally get lean before they try to bulk. The idea is that higher body-fat levels require more fat gain per unit of hypertrophy (i.e., higher body-fat levels impair hypertrophy), so low body-fat levels should be optimal for maximizing hypertrophy in the absence of fat gain. When speculating about why p-ratios should be lower for people with higher body-fat levels, proponents of the idea often lean on two primary explanations: 1) excess body-fat reduces insulin sensitivity, which attenuates hypertrophy by impeding nutrient delivery to muscles; 2) excess body-fat causes chronic elevations of systemic inflammation, which impairs muscle growth.
As discussed in the three-part article series (one, two, three), these mechanistic explanations are unsupported, insufficient, and rarely even quantified in a meaningful way by the people making them. The insulin sensitivity argument has fully collapsed, and has already been abandoned by most people who purport to be evidence-based in their approach to fitness. As I explain here, the inflammation argument is also incompatible with the evidence, and is usually justified by studies showing impaired hypertrophy in mice on high-fat diets (but not other diets), or studies that compare people with higher or lower systemic inflammation levels, while ignoring the fact that the higher-inflammation groups almost never have higher body-fat levels than the lower-inflammation groups. In other words, people are working really hard to try to support this idea, and they lean on research in mice on very specific diets, patients recovering from anorexia, patients transitioning out of “prolonged starvation” protocols, and people (with very average body-fat levels) who are very old or very sick. Or they use fertility as a proxy for muscle hypertrophy – as an outspoken critic of the assumption that muscle protein synthesis is always a suitable proxy for muscle hypertrophy, I probably don’t have to clarify that having a baby is a different thing than making your muscles bigger. This entire approach to the problem seems to rest on the assumptions that 1) nobody has ever studied what happens when human beings lift weights, and 2) even if they did, everyone participating in resistance training studies would have extremely similar body-fat levels.
As it turns out, both assumptions are false. A very large number of studies have assessed hypertrophy in response resistance training, and a wide range of body-fat levels are present within this literature. So, back in 2021, I conducted a participant-level meta-analysis using all of the resistance training data sets I could get my hands on. Broadly speaking, the analysis led to two very intuitive conclusions: 1) baseline body-fat levels don’t meaningfully impact the amount of hypertrophy achieved over the course of a resistance training program; 2) people with higher baseline body-fat levels are more likely to lose some body-fat over the course of a resistance training program. In terms of hypertrophy, we visualized changes in fat-free mass (stratified by sex), and also presented a mixed-sex visualization of direct measures of hypertrophy (for example, ultrasound measurements of muscle thickness). These findings are presented in Figure 1.
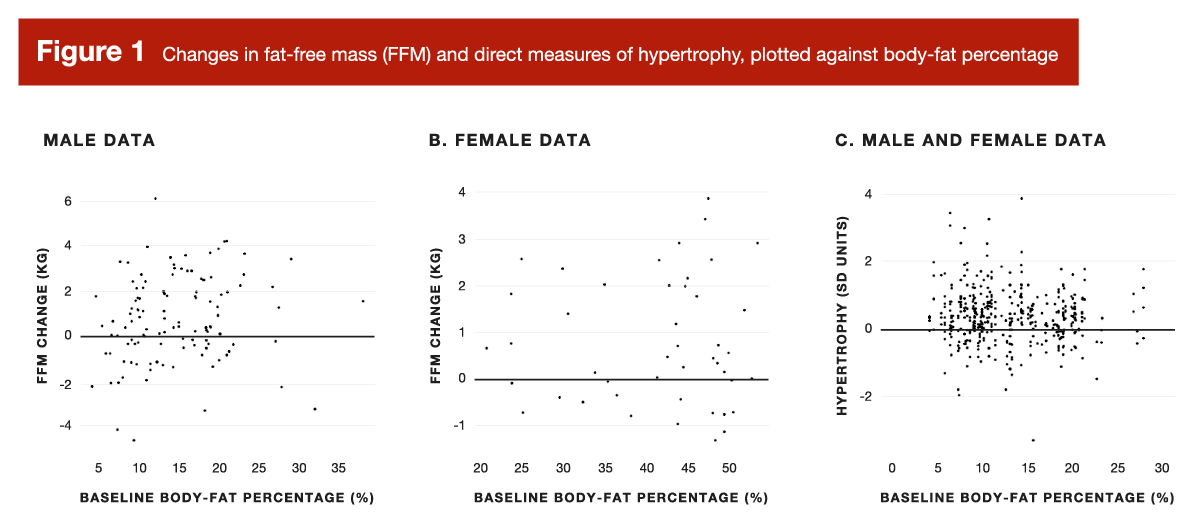
Even without a convoluted statistical explanation, your eyes tell you enough – there’s nothing exciting happening in Figure 1. There was, however, a criticism of our participant-level meta-analysis: some people felt that lower body-fat levels were a bit overrepresented in our data set, and that higher body-fat levels were a bit underrepresented in our data set. In their eyes, this left a bit of hope for applying the p-ratio concept to lifters – perhaps the relationship exists, and is important, but only when body-fat levels are on the higher end of the spectrum.
That’s where the presently reviewed study comes into play (1). In this meta-regression, the researchers sought to “analyze whether the effects of resistance exercise on whole-body fat and lean mass are moderated by exercise dosage (i.e., duration, volume, and intensity), age, body mass index, baseline values, assessment methods, and prescription of aerobic exercise and caloric restriction in overweight/obese adults.” The total sample for this meta-analysis project consisted of 2,537 participants with overweight or obesity, from a total of 85 interventions within 67 separate articles. In other words, this was a huge meta-regression exploring hypertrophy outcomes in a giant sample of people who, on average, had much higher body-fat levels than the leanest people in our previous participant-level analysis. If the previously mentioned criticism was correct, and our analysis was indeed biased by analyzing a sample that was too lean to generalize to individuals with body-fat percentages in the 30s and beyond, then this meta-regression would be perfectly suited to identify that.
In terms of fat loss, the results weren’t particularly shocking. Participants lost an average of 1.6kg of fat, with larger absolute fat losses observed in males when compared to females. Furthermore, fat loss values tended to be greater among participants with higher baseline levels of fat mass. On average, lean mass gains were positive, but modest in magnitude (+0.8kg). In order to determine how certain variables influenced gains in lean mass, there are two ways to look at it. We can use “univariable models,” which are very simplistic. In a univariable model, there are only two variables: one outcome, and one predictor. So, if you constructed a univariable model to determine how age impacts lean mass changes, you would be totally ignoring other important variables (such as intervention duration, weekly volume, and so on) rather than controlling for them. We can also use “multivariable models,” which are more nuanced and comprehensive. In a multivariable model, there is one outcome variable, but several predictors. So, if you constructed a multivariable model to determine how age impacts lean mass changes, you would be looking at the independent impact of age, after adjusting the model to account for all other important variables that you have access to (such as intervention duration, weekly volume, and so on).
Table 1 presents the impact of several key variables, using a combination of univariable and multivariable models. Looking at the univariable results, it appears that age and the magnitude of caloric restriction were predictive of total changes in lean mass throughout the duration of a resistance training intervention (people who were older or more calorically restricted struggled to gain lean mass, which makes sense). The slope for BMI was negative (-0.11), which would suggest that people with higher baseline BMIs found it slightly more challenging to gain lean mass. However, this effect was not statistically significant (p = 0.112). More importantly, this univariable relationship could be meaningfully confounded by other variables. For example, it’s very possible (if not likely) that studies with larger planned caloric deficits would intentionally recruit samples with higher BMI values, because these studies are more explicitly designed to study weight loss. When we look at the multivariable model, this negative slope describing the relationship between BMI and changes in lean mass flips, then doubles (a value of -0.11 changes to +0.22), with a similar p-value (p = 0.112 versus p = 0.106).
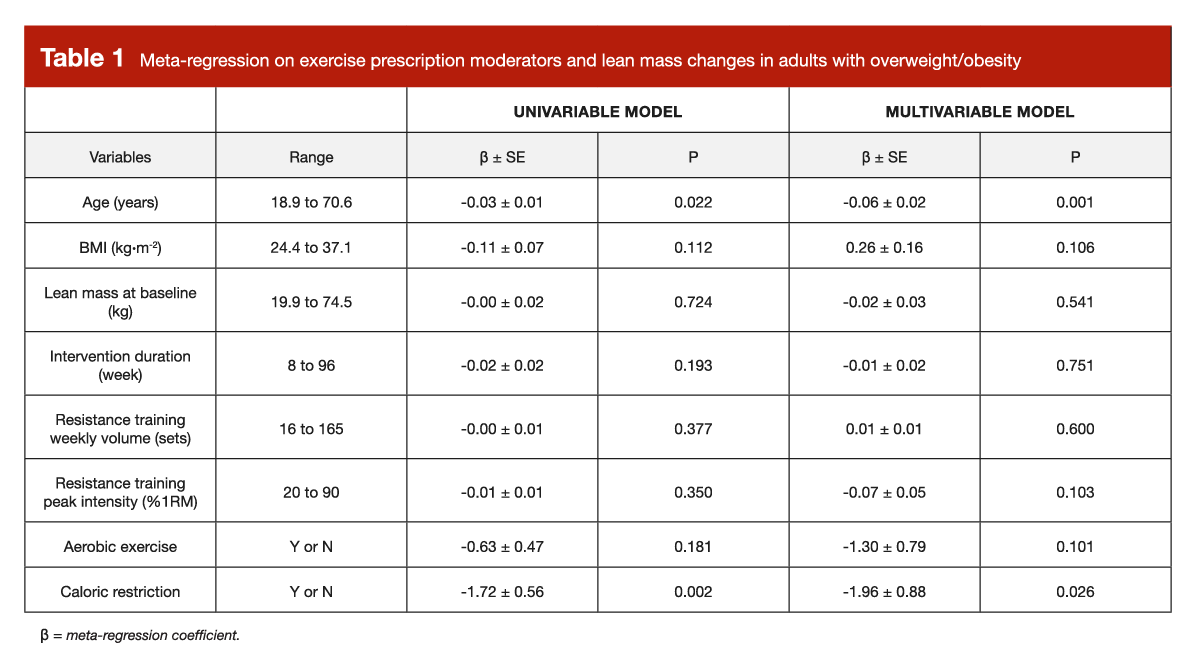
With this in mind, one might argue that baseline BMI (which is a suitable proxy for body-fat level in a population of untrained individuals with overweight or obesity) was unrelated to gains in lean mass. One might argue that higher BMI actually led to better gains in lean mass, after accounting for relevant study characteristics that absolutely should be accounted for. Either way, it would be very, very hard to look at these data and conclude that higher baseline body-fat levels impair hypertrophy to a meaningful degree. One might even argue that these results should be thrown out entirely, given that the absolute magnitude of observed hypertrophy was fairly low. That’s fine, but it still leaves p-ratio proponents with a very inconvenient problem – time and time again, we see studies that absolutely should be able to detect an impairment of hypertrophy induced by high body-fat levels, and the theory fails over and over. If this effect exists, is large enough to care about, and affects people within commonly observed body-fat ranges, it shouldn’t be this hard to find any resistance training studies that are merely compatible with the concept, and it should be way harder to find the many, many resistance training studies that directly contradict the concept.
Looking at the totality of the literature, there is some justification for the p-ratio concept when looking at mice on high-fat diets, patients recovering from anorexia (who aren’t lifting), and patients transitioning out of “prolonged starvation” protocols (who aren’t lifting). However, there is no evidence that we can actually “use” this idea to improve anything at all – the model dictates that leaner people (who aren’t lifting) gain a higher proportion of weight as lean mass, but also that leaner people (who aren’t lifting) lose a higher proportion of weight as lean mass. So, if you wanted to get leaner in order to capitalize on a theoretical boost in gains, you’d have to lose more and more muscle as you went; the hypothetical “prize” of extra gains would just be regaining the muscle you lost when you were trying to get lean enough to boost muscle growth. Putting that consideration aside, the much more important point is that this hypothetical application of the p-ratio concept (that is, the idea that high body-fat levels blunt hypertrophy, or that you can potentiate hypertrophy or access “leaner gains” by getting to a lower body-fat percentage) fails to pan out for old lifters, young lifters, lifters with high body-fat, lifters with low body-fat, trained lifters, untrained lifters, athletes who lift, and non-athletes who lift. The mechanistic justification for applying this concept to lifters is vague, incomplete, and unsupported (one, two, three), and the most relevant literature in human beings lifting weights contradicts the idea time and time again. In short, this popularized application of the p-ratio concept used to be a house of cards that rested on some totally indefensible assumptions, but we can now view it as a scattered pile of cards on a table.
Note: This article was published in partnership with MASS Research Review. Full versions of Research Spotlight breakdowns are originally published in MASS Research Review. Subscribe to MASS to get a monthly publication with breakdowns of recent exercise and nutrition studies.
References
- Lopez P, Radaelli R, Taaffe DR, Galvão DA, Newton RU, Nonemacher ER, et al. Moderators of Resistance Training Effects in Overweight and Obese Adults: A Systematic Review and Meta-analysis. Med Sci Sports Exerc. 2022 Nov 1;54(11):1804–16.
- Forbes GB. Lean Body Mass-Body Fat Interrelationships In Humans. Nutr Rev. 1987 Aug;45(8):225–31.




