Training with tendon injuries can be a frustrating endeavor. Within a session, focus shifts from performance cues to the sensation of pain and impending anxiety regarding what this means for long-term health and training capacity. As a former strength and conditioning coach, as well as someone who has been training consistently for the majority of my life, I have a strong appreciation for the pursuit of strength, and I intimately understand the innate drive to train despite facing injury. Fortunately, I have been able to merge my passion with my knowledge as a Physical Therapist to assist many individuals within this realm. Through practice, I have been able to provide value on an individual level, and I hope to use this platform to offer perspective on a wider scale.
Mandatory disclaimer: This is not meant to be a replacement for a consult with a medical professional. If you are experiencing pain beyond what is deemed normal aches and pains associated with training, you should seek help. A physical therapist can serve an invaluable role in restoring your ability to train following musculoskeletal injuries. However, knowledge and treatment paradigms vary widely between professionals, and tendinopathy management is rapidly evolving with our growing understanding of the disorder, requiring deliberate effort to keep up to date. The best advice I can offer is to do your homework to ensure your therapist is aware of the demands of your chosen sport, remains current in his or her knowledge of tendon rehabilitation, and offers more than passive modalities for “healing.”
This article is going to start from the ground up, going over tendon structure, function, and adaptive capacity, as well as theories of pathology, rehabilitation principles, and ways to maintain a training stimulus while addressing the problem. While I obviously think this is important to gain insight into the “whys” behind the process, I acknowledge not everyone wants to go back to physiology 101. Feel free to skip ahead to the practical recommendations if you want; I did my best to make it a self-contained section. If you’re feeling particularly rushed, you can even jump straight to the summary below and use the remainder of the article as a reference (no judgment).
Anatomy and Biomechanics:
In the simplest sense, tendons are soft, fibrous tissues that connect muscle to bone and function to transmit force to facilitate movement around a joint and absorb external force to limit muscle damage (1, 2). Tendons are predominantly composed of water (approximately 55-70% of total weight) with the remaining dry weight being comprised primarily of collagen (60-85%). Collagen is a stiff structural protein which comes in many variants within humans; however, for our purposes today, we’ll limit the discussion to the two main types found in tendons, Type I and Type III collagen (90% and up to 10% respectively)3. These molecules are organized in a precise, hierarchical fashion parallel to the long axis of the tendon; thus affording the high tensile strength required for their role in transmitting uniaxial forces (2).
Interestingly, the mechanical behavior of a tendon is dependent upon the type of stress applied (4, 5, 40). When exposed to stresses, their ability to stretch and recoil serves a protective role for the attaching musculature, allowing for the delay and reduction of total force (5). Previous studies have demonstrated that under most conditions, stretching of the tendon is out of phase with the adjoining muscle fibers. During eccentric contractions, lengthening of the muscle-tendon unit (MTU) is initially accommodated for almost exclusively by the tendon while the muscle fibers remain the same length (or even shorten). Subsequently, the tendon then recoils as it releases the absorbed energy to skeletal muscle, causing elongation of the contractile fibers. Importantly, the rate at which energy is stored in the tendon is significantly greater than the rate of energy release to the muscle – sparing musculature from high power stressors while prolonging the muscle’s ability to maintain optimal length-tension relationships required for force expression or dissipation.
Additionally, these dynamics change based upon the intensity and rate of the applied load due to a tendon’s viscoelastic characteristics. Like viscous materials, tendons display time-dependent mechanical characteristics in which they become stiffer as the rate of loading increases. This enables the tendon to tolerate a wide bandwidth of velocities with relatively small changes in length between conditions. Like elastic structures, tendons also display time-independent characteristics in which the intensity of the stress drives length changes regardless of the application rate, allowing for greater muscular power (and subsequent movement economy) upon recoil. These two seemingly distinct qualities are present within the same structure in varying proportions based upon the specific task constraints. During the early eccentric contraction, fluid is forcefully pushed out of the tendon, resulting in high viscous resistance. Constant tension (and tissue compression) does not allow for the return of fluid prior to concentric muscular action, leaving the elastic qualities of the tendon to predominate during the concentric phase (4, 40). In totality, this introduces the complex behavior tendons display. Their adaptability is vital to our success as a species, as the ability to buffer stressors of varying magnitude and rate allow for amazing variability in human movement without major adverse responses (4, 5, 6, 40).
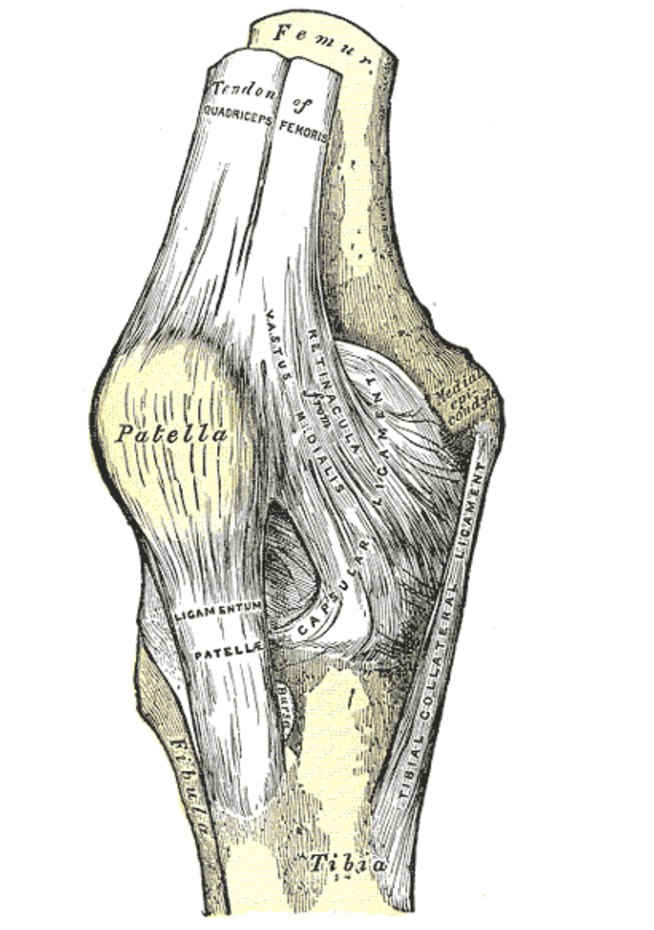
The primary focus of this article is going to be the patellar tendon, due to its propensity for injury within the sport of powerlifting. The patellar tendon attaches to the inferior pole of the patella (knee cap) and the tibial tuberosity (bony protrusion at the top of your shin). Although the majority of the patellar tendon fibers do not directly attach to the quadriceps musculature, they act functionally as a tendon in conjunction with the quadriceps tendon. In this role, the patellar tendon serves to facilitate action of the knee extensors to allow for optimal pull on the tibia during dynamic movement (7).
Scientists estimate that the patellar tendon can withstand between 10,000 to 15,000 N of force, which equates to approximately 13-19x body weight for an 80-kg individual (8). To provide context, Nisell and Ekholm conducted a study where they mathematically estimated patellar tendon loads during the barbell squat (250 kg squat performed by a 110 kg lifter). They demonstrated peak forces only reached 6,000 N – far below the expected failure threshold (9).
Not all tendons serve the same functional role. Tendons are classified into two distinct categories: positional tendons, which act to predominantly position limbs, or energy-storing tendons, which act more as springs to allow for more efficient movement. The patellar tendon is an energy-storing tendon due to its specific functional role and altered physiological properties. In order to effectively store and return energy efficiently, the patellar tendon has higher elastin content, lower absolute levels of collagen, and a higher relative proportion of Type III collagen versus Type I (Type III has lower stiffness, allowing for improved extensibility and recoil) when compared to positional tendons (2). Additionally, energy-storing tendons display significantly lower levels of collagen turnover, resulting in a half-life of approximately 200 years (important concept for rehabilitation purposes to be discussed later).
Adaptability:
Now that some background information has been presented, we can get to the important part: if and how we can alter the physical properties of tendons. Central to this premise is the concept of mechanotransduction. Mechanotransduction, by definition, is the mechanism by which cells convert mechanical stimuli into cellular responses (10). Cells are sensitive to forces such as shear, tension, and compression, and they respond accordingly through cellular proliferation, migration, tissue repair, altered metabolism, and even stem cell differentiation and maturation (10). Thus, in essence, stress is the language our cells speak, serving to stimulate both positive and negative change. Tendon cells are no exception, sensing and responding to stressors in their surrounding environment. Load is a primary stressor which our tendons detect and functions as a vehicle to drive rehabilitative efforts.
The inner portion of a tendon is formed during years of peak growth (during time of puberty) and is essentially not renewed thereafter. Therefore, when exposed to sufficient load, the subsequent changes observed in tendon structure are almost exclusive to the outer periphery of tendons (13).
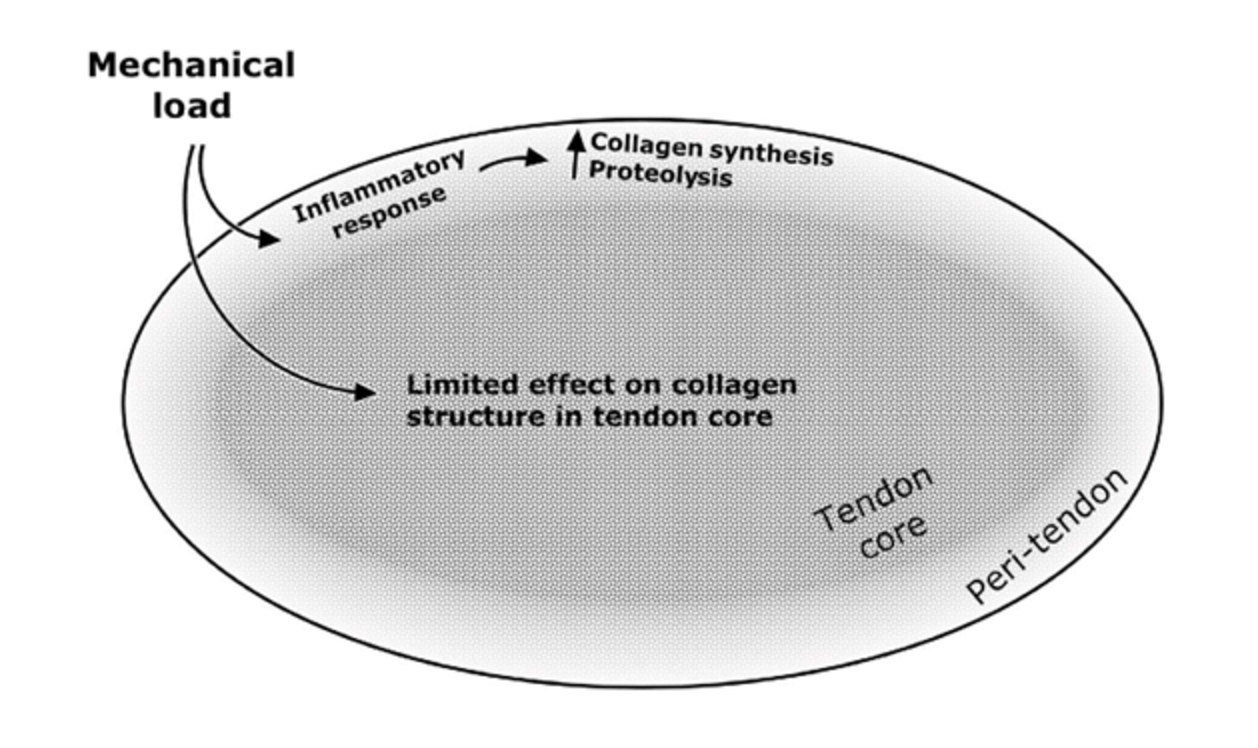
The non-homogenous response to loading contributes to a lower absolute ceiling of adaptability in tendons when compared to musculature. Additionally, in the metabolically active segments of tendons, the tissues also display lower relative adaptive capacity than skeletal muscle due in part to oxygen consumption levels calculated to be approximately 7.5x lower (15). This may paint a bleak picture for our ability to modify tendon structure an appreciable amount, but several recent systematic reviews demonstrate meaningful effects of loading on tendon structure. Primarily, three key features of tendons have been studied in response to acute and chronic loading: tendon mechanical (stiffness), material (Young’s Modulus), and morphological (cross-sectional area, level of molecular cross-linking) properties (11,12). Stiffness refers to the ability to resist deformation, and Young’s modulus refers to stiffness when tendon dimensions are taken into account (12). Alterations in material properties are believed to be the acute response to tendon loading, with significant increases noted within the first several months of training (increased stiffness reportedly ranging from 26% to 85%), and changes in morphological qualities are believed to be the chronic adaptation after years of training (increased cross-sectional area reportedly ranging from 1.5% to 36%) (12).
Tendinopathy
Classically, tendon pathologies were separated into two discrete conditions referred to as tendinitis and tendinosis. Both were considered “overuse” injuries with the former representing an acute, inflammatory condition and the latter representing a degenerative condition. Inflammation’s role in the disorder has been questioned recently, with evidence suggesting that while there are an increased number of inflammatory cells present in pathological tendons, the response observed is not in line with a traditional inflammatory response and the presence of inflammatory markers does not necessarily support inflammation as the primary event or key driver of tendon pathology (16, 17). This revelation, in conjunction with the totality of evidence, led away from the belief that these conditions are mutually exclusive. Therefore, the more generic term “tendinopathy” is preferred over the aforementioned terminology.
Additionally, the causative mechanism of “overuse” is antiquated and perpetuates an inaccurate notion of how biological organisms respond to stress. The amount of stress required to evoke a mal-adaptive tendon response is constantly changing based upon fluctuating individual constraints. For example, an athlete may be able to tolerate a very high workload if they deliberately condition themselves to withstand that level of stress over an appreciable amount of time. That very same athlete may experience negative consequences while performing a fraction of that volume if they are coming off a period of detraining due to the plasticity of various structural, hormonal, and enzymatic qualities which are mediated by habitual loading (3, 10). Hence, we should instead view the triggering event as a ratio instead of an absolute – that the applied loads (magnitude/frequency/rate) exceeded our body’s ability to adequately respond in specific contexts.
Dr. Jill Cook, a leading researcher in the field, proposed that tendinitis and tendinosis are different aspects of the same complex continuum and has organized tendinopathy into three distinct phases: 1) reactive tendinopathy, 2) tendon dysrepair, and 3) degenerative tendinopathy (16, 20). A reactive tendinopathy is believed to be an acute, reversible process brought about by a rapid increase in mechanical loading. The tendon swells due to an increase in water retention and is proposed to be a protective response to reduce stress along involved collagen fibers (20, 21). Tendon dysrepair is suggested to follow if loading exceeds tendon capacity for a substantial period of time. Here, we continue to see increased fiber diameter, but the tendon matrix begins to breakdown and there is evidence of neural and vascular ingrowth. Finally, a tendon reaches a degenerative state, characterized by further collagen disorganization, advanced matrix breakdown, and increased fiber thickness.
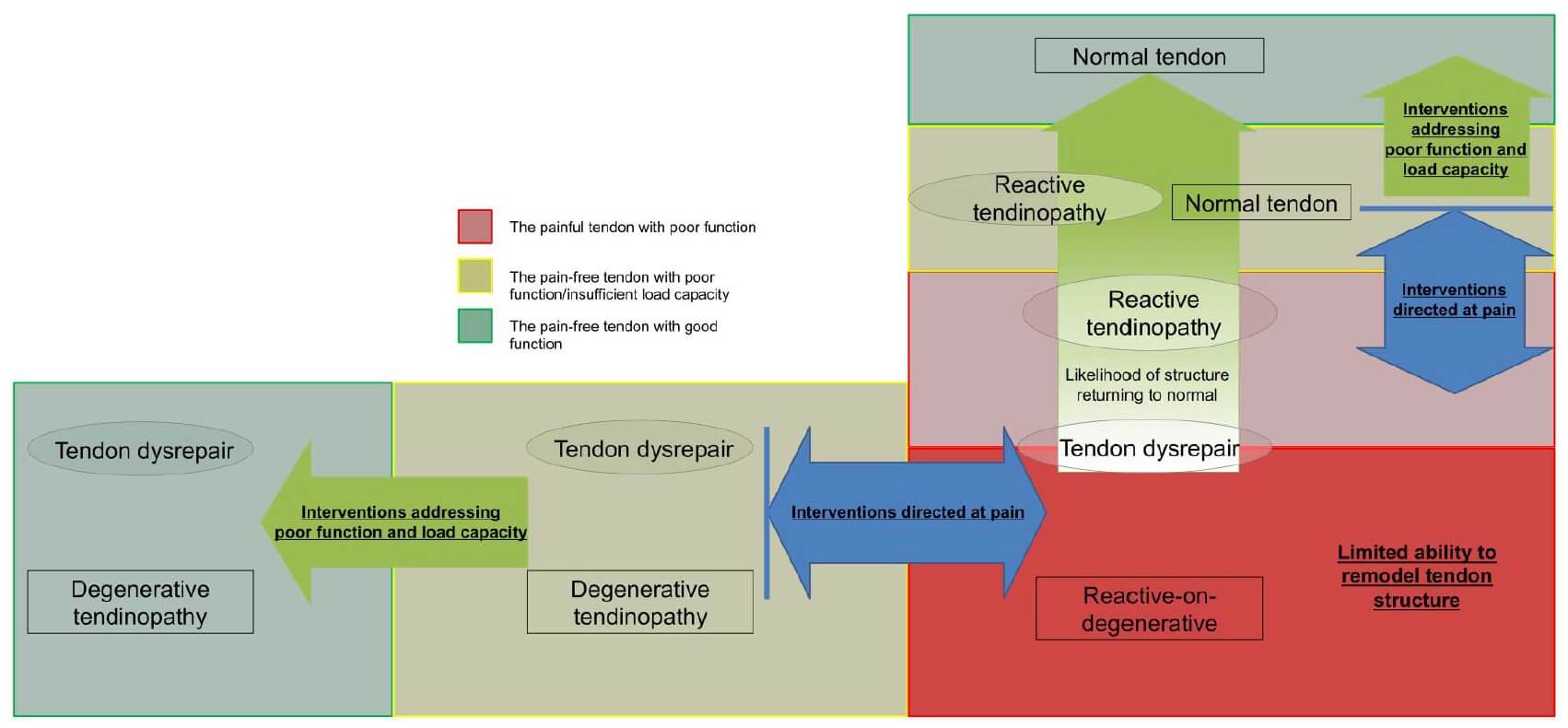
In clinical findings, the hallmark features of patellar tendinopathy across all three stages include pain localized to the inferior pole of the patella and load related pain that increases with the demand on the knee extensors. To differentiate a tendon disorder from other sources of similar symptoms, it should be noted that there is rarely pain in resting states and we typically see a dose-dependency of pain, with increased intensity or rate of loading associated with increased reports of pain (18). Again, I am not condoning self-diagnosis of the disorder. If your condition aligns with these symptoms, or there is any pain beyond normal aches associated with training, I would seek a medical provider.
Practical Implications
Tendon Rehab
The most important concept to understand with tendon rehabilitation is that there is very little evidence to suggest there is any reversibility noted within affected regions of degenerative tendons – with minimal physiological adaptations noted following rehabilitative efforts (16, 21, 22). At first, this would appear to contradict the adaptive process detailed above. But, once the extracellular matrix is disrupted beyond a certain threshold, the involved region loses its ability to transmit load; thus, there is no mechanical stimulus to force change (22). However, because there is enough redundancy within tendons, we do not need this to occur specifically at the site of degeneration. A recent ultrasound study demonstrated that pathological tendons have an increased mean cross-sectional area and an increased anterior to posterior diameter compared to non-pathological tendons. And while there is a reduced relative percentage of aligned fibrillar structures (and a coinciding increase in the percentage of disorganized tissues), in absolute terms, these tendons contain increased amounts of load-bearing tissues comparatively. Therefore, while our efforts may not impact areas of disorganization within an involved tendon, we can evoke desired changes within the plentiful areas of aligned collagen. This idea can be adequately visualized by the metaphor “treat the donut, not the hole” where the “donut” refers to the viable collagen structures and the “hole” refers to the degenerative and/or disorganized fibers (16).
First and foremost, we are concerned with optimal tissue loading to facilitate the desired response. There is a pervasive notion that more is better. However, as mentioned above, loading beyond our ability to adapt will have detrimental consequences and will only serve to perpetuate our cycle of frustration. Dr. Scott Dye proposed a tissue homeostasis model which included a concept known as the “envelope of function,” serving to demarcate the boundaries for the safe range of load acceptance and transference within a given period of time (23). To force positive adaptations, we must find the sweet spot within the envelope of function where the total stress is enough to disrupt tissue homeostasis and create an impetus for change without diminishing structural integrity.
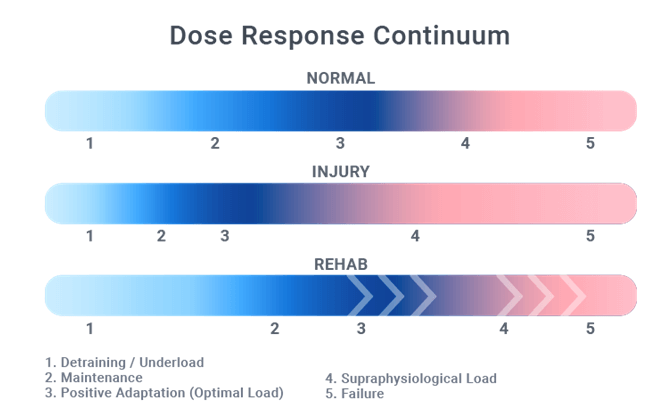
So the important question is how do we know how much is right? Research groups studying tendon rehabilitation have adopted a system based on subjective feedback regarding pain within training and over a 24-hour window immediately following (18).
Important tangent: Pain is a widely misunderstood phenomenon, with many misinterpreting pain as a direct indication of tissue damage. While pain neuroscience is beyond the scope of this article, it is imperative to understand that pain is a protective mechanism of the body and not simply a reflection of the tissue state. While pain does indeed strongly correlate to tissue damage in acute traumatic events, this association is diminished over time, and tissue physiology does not predict pain response in chronic conditions. Therefore, do not panic if you experience low levels of pain, you are (probably) not doing significant harm and you are (probably) not experiencing a major setback. (For more on the topic of pain, please read this free full text from Lorimer Moseley)
Typically, you will be able to determine tissue tolerance to load based upon a simple self-assessment within a specific movement pattern. (This will look familiar to those of you who are aware of David Dellanave’s Biofeedback training. Similar concept utilized for rehab purposes.)
- Find a movement that you can perform consistently to stress the patellar tendon. In research, this is often a single leg squat performed on a decline board; however, all that matters is that you choose a task which directly stresses the tendon and can be performed in a consistent manner on a daily basis.
- Note your level of pain within this task on a 0-10 scale and use this as your reference point.
- Perform your training session while modifying stress (through any of the variables to be discussed soon) as to maintain, at most, low-intensity pain. The threshold is variable depending on the individual, but I typically stop patients between 3-4 out of 10.
- Check your chosen pattern at the same time of day, every day, and log your level of discomfort. If your pain is below or remains the same as your initial assessment: You are probably tolerating the current level of stress well and can maintain or increase the training demands. If your pain increases compared to your initial assessment: The total stress may be too much and you may need to alter your training strategy.
Training Considerations:
Each of the upcoming variables impact total stress upon the involved tendon in varying capacities. Finding the right balance while ensuring you are addressing qualities you wish to develop is a difficult process. The best I can offer is education on several factors believed to be important. I will leave the nuts and bolts of programming up to you on an individual basis.
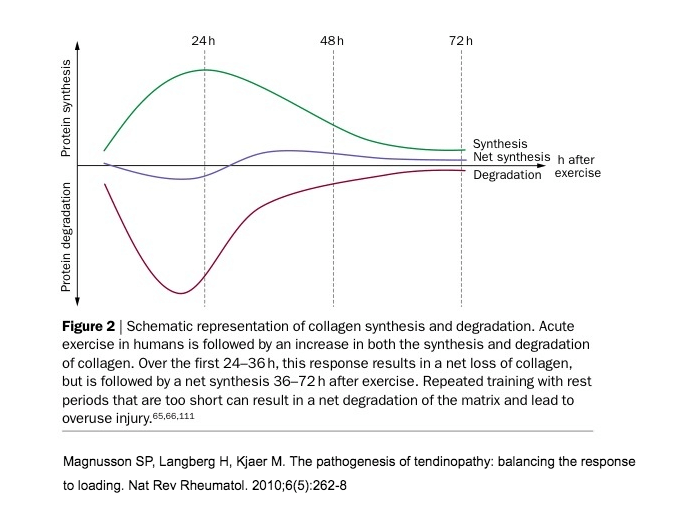
In order to improve load-bearing capacity of the viable structures within pathological tendons, we need to ensure net synthesis of collagen. Following resistance training, there is an increase in collagen turnover, both synthesis and degradation. Initially, enzymatic activity leads to a net protein loss, with a net gain not observed until approximately 36 hours after exercise (see figure below). Therefore, it is believed that for positive adaptations to be observed, a certain restitution period is beneficial. Without adequate recovery time, it is likely there will be a net loss of collagen, rendering the tendon vulnerable to excessive loading (3). Considering these findings, it is best to space training sessions out by at least 36-48 hours to facilitate desired outcomes. Although I make this recommendation, there are many studies which have used daily low-volume loading programs successfully (39). These outcomes mean there is likely a threshold effect for the collagen turnover rates discussed and the extended periods of reduced loading may not be required for positive results. But for practical purposes, the suggested training frequency is sufficient and convenient for most.
Training Volume
The careful administration of training volume is critical to providing optimal levels of stress. Greg has done an excellent job of discussing the merits of different volume metrics here, but for our purposes, any method which can be performed consistently will suffice. We must find and implement a training volume within our envelope of function, somewhere between the minimum effective dosage and our maximum recoverable volume (MRV). While it may sound appealing to jump to our MRV, this presents several challenges in the rehabilitative process.
The stress applied to tendons is not isolated within the four walls of the gym and this concept demands particular consideration for tendons of the lower body, such as the patellar tendon, which are required for locomotion. Stresses associated with daily demands typically represent a small absolute value of our tendon’s capacity. However, in pathological tendons, these same stressors represent a significantly higher relative percentage of that capacity and may have a critical impact upon your ability to train. The daily fluctuation in additional load is hard to quantify and account for, rendering it difficult to walk the fine line of your MRV within session. Additionally, the magnitude of difference in adaptations observed between performing the MRV and those seen using a more conservative volume is speculatively not large enough to overcome the detriment of overshooting your tendon’s current capabilities.
This leads to the notion that it is much more reasonable to underestimate yourself at first, ensure you are tolerating the current load well (using the 24-hour symptom response noted above), and gradually build volume accordingly.
Exercise Selection and Order
To this point, an unmentioned consideration is the impact a condition such as tendinopathy has on neural regulation of force expression. Paradoxically, individuals with tendinopathy display both elevated corticospinal excitability as well as cortical inhibition of the quadriceps musculature (24, 25, 26). It is believed there is greater pre-activation and enhanced excitability of the quadriceps in order to increase muscular stiffness to accommodate for the greater compliance of pathological tendons, with increased cortical inhibition postulated to limit total force capacity (24, 25, 32). While these neural abnormalities may be inefficient (likened to a novice driver controlling the speed of their car with one foot on the brake and one on the accelerator) they are protective in nature, serving to reduce mechanical loading of the vulnerable tissues (26). Obviously, this regulatory strategy may impact our ability to load the involved musculotendinous unit as desired, and is clearly not ideal for anyone wishing to maximize muscular hypertrophy or improve their powerlifting total. The fortunate news is that recent investigations have indicated we may be able to significantly alter the aforementioned abnormalities through relatively simple means. Rio et al. conducted a study on patients with patellar tendinopathy where they compared the influence of isometric contractions to isotonic contractions on pain, quadriceps strength (measured in terms of maximum voluntary isometric contractions (MVIC)) and measures of corticospinal inhibition (25). Performing five 45 second isometric knee extensions (60 degrees of knee flexion) with 70% 1RM had significantly greater impact on all three variables than the isotonic exercise. More specifically, the reduced inhibition achieved through isometric loading led to an average MVIC increase of 18.7% immediately following the intervention, with sustained improvements noted for at least 45 minutes. Note: This does not imply you will squat 20% more during your training. It does, however, provide preliminary data to support the use of moderate-to-high intensity isometric loading to start your training session. This will assist in normalizing the neuromuscular control of the involved lower extremity and potentially improve load tolerance and force expression for the duration of your routine.
(The Spanish Squat is a commonly substituted exercise if you don’t have a leg extension machine available to you).
Following a warm-up period which includes isometric loading, it is time to choose a primary squat pattern. Greg has extensively written in regards to the difference between squat techniques when it comes to probable muscular effort and development. And while the discrepancies in muscular demands are likely small, the movement pattern chosen can have significant impact on the stress placed upon the patellar tendon. Mathematical models have demonstrated patellar tendon force (through a combination of tensile and compressive stress) is directly proportional to knee flexion angle during the squat (8). Stress is maximized at angles of deep knee flexion (reaching approximately 6000 N at 130 degrees) and slowly decreases to 2000 N by 30 degrees. Therefore, particularly during early stages of rehabilitative efforts, it is wise to limit loading in significant knee flexion (18, 27, 28). Practically, this leads to choosing options where your movement is constrained biomechanically rather than consciously adjusting your depth or effort. A great starting point would be a box squat. Within this pattern, you are able to sit back an unnatural amount to increase the hip flexion moment (thus increasing hip extension demands) while maintaining a shallow, vertical tibia. Additionally, a box affords you the specific control over knee angles which allows you to train at or below your chosen low-intensity pain threshold. Over time (weeks to months), a simple progression would be to move to a low-bar back squat, followed by high-bar back squat, and finally front squat (if so desired) as tissue tolerance allows.

Photo from: http://www.thebarbellphysio.com/squat-assessment-checklist/
Great resource for clinicians working with athletic populations.
In conjunction with modifying your squat selection, the benefit of accommodating resistance should be intuitive. The ability to progressively load the squat as you rise from the bottom position can be useful to maximize the training effect without excessively loading tendons in their most susceptible position. When controlling for other variables (such as intensity and velocity), it has been demonstrated that forces on the patellar tendon during the eccentric portion of a squat are 15-35% greater than the concentric (8). Thus, chains or a reverse-band configuration are probably good options here for altering the resistance throughout the movement.
Finally, it is commonly hypothesized that solely performing compound movements may not adequately stress the involved tendon due to conscious or subconscious compensatory strategies (18). This may be as blatant as a deviated movement pattern to shield sensitized tissues or could be as insidious as the neural protective adaptations outlined above. Therefore, it is probably best to include some form of isolated leg extension towards the end of your training session to ensure adequate loading of the involved musculotendinous unit. Be sure to account for this when determining your appropriate training volume
Training Intensity
Traditionally, eccentric contractions were thought to be the vital component of tendon rehab and were used extensively to address the pathological condition. However, Bohm et al. recently conducted a systematic review where they examined the influence of different loading conditions and types on adaptive response. They found that type of contraction used is irrelevant (with one notable exception to be mentioned later), and the primary difference in outcomes was attributable to the intensity of exercise used (11). So, the natural question is what is the optimal intensity to drive tendon adaptations? To clarify, by intensity, I do not mean effort. In this context, intensity implies percentage of your one repetition maximum. While skeletal muscle is extremely responsive to a wide variety of stimuli, with positive adaptations noted in a multitude of training conditions, tendons require more specific loading parameters11,29,30. When controlling for effort, muscular adaptations have been demonstrated between low and high load conditions (between 30-90% 1RM), whereas tendons do not express significant mechanical, material, or morphological adaptations when loaded below 70% 1RM (11, 29). Higher intensity contractions are hypothesized to induce greater strain on the involved tendon, leading to a loss of normal collagen crimp, increased fiber recruitment, and greater cell deformation – a necessary stimulus to evoke the intended consequences (11). Therefore, it is important to perform your targeted exercises at or above 70% 1RM to elicit desired changes.
(70% 1RM usually correlates to your 10-15 rep max; however, this number is highly variable between individuals and can differ from exercise to exercise. It is best to estimate your 1RM by performing a rep max test with the highest intensity you can without experiencing pain).
Training Velocity
The exception to the irrelevance of contraction type on tissue adaptation is plyometric contractions. Load duration appears to be an important consideration when determining exercise selection. The incorporation of heavy, slow resistance training has grown in popularity due to the positive outcomes achieved, whereas rehab efforts solely using high velocity, plyometric contractions have failed to consistently produce significant tendon adaptations (11).
Speculatively, high loading rates have also commonly been viewed as a risk factor for tendon damage and have been proposed to be an important modifiable variable to consider when performing resistance training (7, 8, 18). Fortunately, Earp et al. conducted a study using barbell back squats to gain insight on tendon dynamics (and subsequent stress) when velocity demands are changed. They had participants perform back squats using 60% 1RM either using a deliberate tempo (2 s eccentric, 1 s pause, 2 s concentric), a self-selected pace without a pause, or perform a maximum-speed jump squat. Viscous qualities of the tendon unit allowed for very little difference in tendon lengthening early in the eccentric contraction despite large fluctuations in speed. However, rate of tendon loading was greater in the high-velocity conditions during this period – leading to greater force through the tendon during the late eccentric phase and early concentric phase comparatively (40). This knee angle, as noted above, is the most mechanically vulnerable position for the tendon and high levels of stress within this specific posture pose a substantial concern for overloading a pathological tendon. Based on this knowledge, it may initially seem appealing to remove all high-velocity training from your program. However; this should come as a tough pill to swallow, considering recent evidence suggests training at your maximum velocity is a valuable stimulus for improved muscular strength and hypertrophy. Luckily, a closer look provides room for optimism.
An important distinction is the difference between intent to move at maximum velocity versus actually moving at a high velocity. It has been proposed that intention to move at maximum speed, rather than the specifics of the movement itself, is the critical factor to the muscular adaptations discussed above (33). And while this concept is far from unequivocally accepted, there is enough evidence to suggest effort is a major contributor in this response through neurophysiological mechanisms. Unlike muscle tissue, the discrepancies in stress to the tendons appear highly specific to the velocities rather than intent. Thus, if we choose the right parameters for exercise (primarily heavy enough load), we can constrain the absolute velocity of the squat (to limit tendon stress) while still providing maximal effort to move as quickly as possible (to elicit muscular adaptations). Additionally, even if we were to train at higher movement speeds, the stress experienced within the tendon changes throughout the squat. In Earp’s research, once participants initiated concentric contractions, rate of force development through the tendon drops to a fraction of the values obtained during the late eccentric phase. Furthermore, the absolute values of tendon force were significantly lower during the concentric portion of the lift compared to eccentric at all velocities measured (40). This finding indicates that velocity’s contribution to stress is amplified during the eccentric phase of the squat and mitigated during the concentric.
Earp’s research, taken together with literature concerning intent’s role in muscular adaptations, provides evidence to the notion that modifying velocity during the eccentric portion of the squat is beneficial to limit total stress, while it is probably unnecessary, and potentially suboptimal, during the concentric aspect. Therefore, it is recommended to use a slow, deliberate tempo during the lowering phase (typically 3-4 second) followed by maximum effort concentrically.
You can simply perform a self-timed descent, or you can implement an external pacing device such as a metronome. The choice may matter, as preliminary data has revealed an interesting observation from performing tempo training with auditory cueing. Imaging studies have shown that there are different activation patterns in the brain, with broad cortical and subcortical activation seen in self-paced movements compared to highly localized cortical activation when performing metronome-paced movement (34). This brain activity manifests as changes in excitatory and inhibitory neural activity similar to that observed following prolonged isometric contractions, leading to the common adoption of metronome training to address tendinopathies (24, 34). However, it is important to note that this data is in its infancy: The neural response has not been investigated within the specific context of patellar tendinopathies, the movements studied are typically simple, single-joint contractions, and it is yet to be determined if there is an additive effect when combined with isometric loading. The changes observed may simply be due to an external focus during the movement task and there is probably nothing specific regarding metronome training that is vital to obtaining this result. Pragmatically, however, the effort required to use an external pacing device is small, leaving you with a favorable risk-reward ratio regardless of the true effectiveness.
Rehab Duration
The final consideration is the duration of time you should be actively attempting to modify training stress to accommodate for the injured tissue. In short, you probably always want to monitor total training load to some extent when considering the known injury risk when acute workload drastically differs from chronic levels (35). However, the most important time frame for careful monitoring is within the first 8 to 12 weeks from the start of rehabilitation efforts. The literary consensus is that improvements in tendon pain and function are not correlated to changes in tendon structure (36). Pain, as alluded to earlier, is a complex phenomenon that relays information regarding more than simply the state of the peripheral tissue. The noxious sensation associated with tendinopathies is usually reduced significantly within the first several weeks after introducing therapeutic interventions; however, this does not mean your time rehabbing your injury has concluded. While there may a drastic reduction in sensitivity, tendon material properties are relatively unchanged until approximately the 8-12 week mark and morphological adaptations likely take much longer (currently, no definite time-course for adaptation has been agreed upon but consistent findings appear after years of training) (11, 12). Prospective studies have demonstrated that underlying structural pathology increases the risk of developing symptoms; thus, the patellar tendon is particularly susceptible within the crucial period of time where most athletes feel pain-free despite the persistence of structural insufficiencies (16).
Additionally, the potential risks are amplified when considering the mismatch between muscle and tendon development. Muscular adaptations are noted as early as 3-7 weeks, and muscular force generation capacity is largely influenced by neural mechanisms early in resistance training programs. In contrast, tendons rely predominantly on structural factors for their strength and their physical capacity is minimally influenced until anatomical adaptations occur (37, 38). Thus, within the first several months of rehab, the pathological tendon may be subjected to significantly greater levels of stress from the quadriceps musculature than it is capable of tolerating (31). The allure of lifting at your previous level is hard to suppress, especially when under the guise of full-health due to a pain-free state. Fight the urge and trust the process. Patience is a virtue when returning from an injury, and even if you do everything right, you can’t rush physiology. Therefore, deliberate efforts should be made to account for total stress within the first three months independent of the presence of pain, and progress should be made in conservative increments.
In Summary:
- If there is pain, it is advised you seek medical consult. Even if you hold an adequate knowledge of tendon rehabilitation, there are many additional considerations which need to be addressed. Mobility and strength deficits above and below the knee are commonly observed following prolonged patellar tendinopathy, and many display maladaptive compensatory loading strategies which can contribute to future ailments. Expert evaluation is warranted in most situations.
- Stress is the language our tendons speak. We must stress the involved structures (through deliberate loading) within our individual envelope of function to facilitate desired adaptations.
- The degenerative portion of tendons display little to no adaptations following progressive loading…and this is ok. Pathological tendons compensate for areas of disorganization by increasing the absolute amount of aligned collagen fibers (those capable of transmitting force), and this should be the focus of rehab efforts. “Treat the donut, not the hole”
- Use a within-session and post-session subjective monitoring system to ensure load tolerance. Keep within-session discomfort to at or below 3-4/10 and ensure daily testing remains at or below the initial level.
- Begin training sessions with moderate to high intensity isometric contractions to desensitize the involved tissues and reduce cortical inhibition (typically 5×45 seconds, 70% 1RM leg extension performed between 30-60 degrees of knee flexion).
- Initially avoid loading into deep knee flexion angles. Progress from box squats to low-bar back squats, to high-bar back squats, to front squats (if wanted). The use of accommodating resistance can be beneficial to reduce absolute loads in the bottom position of a squat.
- Finish training sessions with isolated leg extensions (machine or Spanish squat) to address potential stress shielding of the involved tendon.
- Volume must be consistently tracked. Err on the conservative side, staying closer to your minimal effective dose rather than your maximum recoverable volume, and gradually build session and weekly volume as tolerance allows.
- Include high-intensity exercises, at or above 70% 1RM, to adequately drive the remodeling of collagen structures.
- Perform all major lifts with a slow, deliberate eccentric (preferably with an external auditory cue) and maximum effort concentric.
- Ideally, allow 36-48 hours between training sessions to optimize net collagen synthesis.
- Rehab efforts must be consistent for at least 8-12 weeks. Tendon metabolic rate is slower and the response is generally smaller when compared to activity seen in the adjoining musculature. One must account for this for several months while the tendon is still in a vulnerable state.
For anyone looking to keep up to date with tendon rehab, I highly recommend following Peter Malliaras’ blog on http://tendinopathyrehab.com/. Many of his posts were valuable resources for this article.
Read Next
- Benefits of Static Stretching Stretched out of Proportion?
- The Best Drills for Healthy Shoulders and a Big Bench
- How to Prevent Muscle Strains
Share this on Facebook & join in the conversation
References
- Sharma P, Maffulli N. Tendon injury and tendinopathy: healing and repair. J Bone Joint Surg Am. 2005;87(1):187-202
- Thorpe CT, Screen HR. Tendon Structure and Composition. Adv Exp Med Biol. 2016;920:3-10
- Magnusson SP, Langberg H, Kjaer M. The pathogenesis of tendinopathy: balancing the response to loading. Nat Rev Rheumatol. 2010;6(5):262-8
- Pioletti DP, Rakotomanana LR, Benvenuti JF, Leyvraz PF. Viscoelastic constitutive law in large deformations: application to human knee ligaments and tendons. J Biomech. 1998;31(8):753-7
- Roberts TJ, Konow N. How tendons buffer energy dissipation by muscle. Exerc Sport Sci Rev. 2013;41(4):186-93
- Kösters A, Wiesinger HP, Bojsen-møller J, Müller E, Seynnes OR. Influence of loading rate on patellar tendon mechanical properties in vivo. Clin Biomech (Bristol, Avon). 2014;29(3):323-9
- Schoenfeld BJ. Squatting kinematics and kinetics and their application to exercise performance. J Strength Cond Res. 2010;24(12):3497-506
- Escamilla, R. F. (2001). Knee biomechanics of the dynamic squat exercise. Medicine and science in sports and exercise, 33(1), 127-141
- Nisell R, and Ekholm J. Joint load during the parallel squat in powerlifting and force analysis of in vivo bilateral quadriceps tendon rupture. Scandinavian Journal of Sports Sciences. 8: 63 – 70. 1986
- Dunn SL, Olmedo ML. Mechanotransduction: Relevance to Physical Therapist Practice-Understanding Our Ability to Affect Genetic Expression Through Mechanical Forces. Phys Ther. 2016;96(5):712-21
- Bohm S, Mersmann F, Arampatzis A. Human tendon adaptation in response to mechanical loading: a systematic review and meta-analysis of exercise intervention studies on healthy adults. Sports Med Open. 2015;1(1):7
- Wiesinger HP, Kösters A, Müller E, Seynnes OR. Effects of Increased Loading on In Vivo Tendon Properties: A Systematic Review. Med Sci Sports Exerc. 2015;47(9):1885-95
- Heinemeier KM, Schjerling P, Heinemeier J, Magnusson SP, Kjaer M. Lack of tissue renewal in human adult Achilles tendon is revealed by nuclear bomb (14)C. FASEB J. 2013;27(5):2074-9
- Kjaer M, Bayer ML, Eliasson P, Heinemeier KM. What is the impact of inflammation on the critical interplay between mechanical signaling and biochemical changes in tendon matrix?. J Appl Physiol. 2013;115(6):879-83
- Abate M, Silbernagel KG, Siljeholm C, et al. Pathogenesis of tendinopathies: inflammation or degeneration?. Arthritis Res Ther. 2009;11(3):235
- Cook JL, Rio E, Purdam CR, Docking SI. Revisiting the continuum model of tendon pathology: what is its merit in clinical practice and research?. Br J Sports Med. 2016
- Dean BJ, Gettings P, Dakin SG, Carr AJ. Are inflammatory cells increased in painful human tendinopathy? A systematic review. Br J Sports Med. 2016;50(4):216-20.)
- Patellar tendinopathy: clinical diagnosis, load management, and advice for challenging case presentations (Malliaras P, Cook J, Purdam C, Rio E. Patellar Tendinopathy: Clinical Diagnosis, Load Management, and Advice for Challenging Case Presentations. J Orthop Sports Phys Ther. 2015;45(11):887-98
- Bass E. Tendinopathy: Why the Difference Between Tendinitis and Tendinosis Matters. International Journal of Therapeutic Massage & Bodywork. 2012;5(1):14-17
- Cook JL, Purdam CR. Is tendon pathology a continuum? A pathology model to explain the clinical presentation of load-induced tendinopathy. Br J Sports Med. 2009;43(6):409-16
- Docking SI, Cook J. Pathological tendons maintain sufficient aligned fibrillar structure on ultrasound tissue characterization (UTC). Scand J Med Sci Sports. 2016;26(6):675-83
- Malliaras P, Purdam C, Maffulli N, Cook J. Temporal sequence of greyscale ultrasound changes and their relationship with neovascularity and pain in the patellar tendon. Br J Sports Med. 2010;44(13):944-
- Dye SF. The knee as a biologic transmission with an envelope of function: a theory. Clin Orthop Relat Res. 1996 Apr;(325):10-8.
- Rio E, Kidgell D, Moseley GL, et al. Tendon neuroplastic training: changing the way we think about tendon rehabilitation: a narrative review. Br J Sports Med. 2016;50(4):209-15
- Rio E, Kidgell D, Purdam C, et al. Isometric exercise induces analgesia and reduces inhibition in patellar tendinopathy. Br J Sports Med. 2015;49(19):1277-83
- Rio E, Kidgell D, Moseley GL, Cook J. Elevated corticospinal excitability in patellar tendinopathy compared with other anterior knee pain or no pain. Scand J Med Sci Sports. 2016;26(9):1072-9
- Cook JL, Purdam C. Is compressive load a factor in the development of tendinopathy?. Br J Sports Med. 2012;46(3):163-8
- Rudavsky A, Cook J. Physiotherapy management of patellar tendinopathy (jumper’s knee). J Physiother. 2014;60(3):122-9
- Morton RW, Oikawa SY, Wavell CG, et al. Neither load nor systemic hormones determine resistance training-mediated hypertrophy or strength gains in resistance-trained young men. J Appl Physiol. 2016;121(1):129-38
- Cormie P, Mcguigan MR, Newton RU. Developing maximal neuromuscular power: Part 1–biological basis of maximal power production. Sports Med. 2011;41(1):17-38
- Mersmann F, Bohm S, Schroll A, Marzilger R, Arampatzis A. Athletic training affects the uniformity of muscle and tendon adaptation during adolescence. J Appl Physiol. 2016;121(4):893-899
- Chang YJ, Kulig K. The neuromechanical adaptations to Achilles tendinosis. J Physiol (Lond). 2015;593(15):3373-87
- Behm DG, Sale DG. Intended rather than actual movement velocity determines velocity-specific training response. J Appl Physiol. 1993;74(1):359-68
- Leung M, Rantalainen T, Teo WP, Kidgell D. Motor cortex excitability is not differentially modulated following skill and strength training. Neuroscience. 2015;305:99-108
- Hulin BT, Gabbett TJ, Lawson DW, Caputi P, Sampson JA. The acute:chronic workload ratio predicts injury: high chronic workload may decrease injury risk in elite rugby league players. Br J Sports Med. 2016;50(4):231-6
- Drew, B. T., Smith, T. O., Littlewood, C., & Sturrock, B. (2012). Do structural changes (eg, collagen/matrix) explain the response to therapeutic exercises in tendinopathy: A systematic review. British Journal of Sports Medicine, 48(12), 966–972. doi:10.1136/bjsports-2012-091285
- Seynnes OR, De boer M, Narici MV. Early skeletal muscle hypertrophy and architectural changes in response to high-intensity resistance training. J Appl Physiol. 2007;102(1):368-73
- Goreham C, Green HJ, Ball-burnett M, Ranney D. High-resistance training and muscle metabolism during prolonged exercise. Am J Physiol. 1999;276(3 Pt 1):E489-96
- Rutland M, O’Connell D, Brismée J-M, Sizer P, Apte G, O’Connell J. EVIDENCE–SUPPORTED REHABILITATION OF PATELLAR TENDINOPATHY. North American Journal of Sports Physical Therapy : NAJSPT. 2010;5(3):166-178.
- Earp JE, Newton RU, Cormie P, Blazevich AJ. Faster Movement Speed Results in Greater Tendon Strain during the Loaded Squat Exercise. Front Physiol. 2016;7:366