In previous decades, there has been no shortage of inaccurate and misleading messages surrounding meal timing and the importance of when we eat. To name just a few:
- You should eat breakfast to “boost your metabolism.”
- Eating carbohydrates after 6pm will lead to immediate fat gain.
- Eating several smaller meals across the day is required to ensure a “fast metabolism.”
- Nutrient timing is THE critical nutrition factor in sports nutrition.
- Immediate and precise post-workout nutrition is vital for attaining the benefit of your training session.
You have likely come across these flawed soundbites (or similar ideas) at some point in the past. Thankfully, these days, there seems to be an increased understanding of the inaccuracies of such statements and the potential trap of investing too heavily in the importance of meal/nutrient timing. The problem, though, is that this can lead to overcompensation and the perspective that once overall daily calories and macronutrients are equal, then nutritional factors like meal timing, length of the feeding window, and the distribution of those calories/nutrients across the day aren’t worth worrying about (especially in relation to body composition). This leads people to think “when we eat doesn’t matter.” Not only is this nihilistic rhetoric not correct, it could cause people to adopt eating behaviors that potentially fly in the face of what would lead to improved long-term metabolic health and possibly better body composition.
In fact, a sizable amount of research is mounting to suggest that there are very real and important implications for when we eat. The cornerstone for many of the underlying mechanisms relates to impacts on, and impacts of, the circadian rhythmicity of many processes in the body. In this article, we will explore this intersection of circadian biology and diet (termed “chrononutrition”). First, we must lay some foundation for that discussion by understanding what exactly circadian biology is and why circadian rhythms are so important to our health, performance, and body composition.
Cycles in Biology
Cycles of opposing processes are a common aspect of biology. Think of anabolic and catabolic processes. As readers of this site, you will be familiar with muscle protein, for example, where we have muscle protein synthesis and muscle protein breakdown. Despite our fantasies of keeping MPS elevated forever with zero MPB, we know that not only is this impossible, but we actually need both of these processes. Protein breakdown is an important part of the process of remodeling muscle protein. So, for optimal muscle function, it isn’t that one process is “better” than the other, but rather they are both important and we want to cycle back and forth between more/less of each. The goal then is to have appropriate amounts of each and increase or decrease them at appropriate times.
Driving a car is a useful analogy for viewing cycles of opposing processes. The accelerator and brake have opposing functions. One is not inherently “better” than the other. Rather, using each at the appropriate time and for the appropriate duration is what matters. Also, we don’t put our foot down on both at the same time. It’s important that we have these ordered sequences of biological rhythms for similar reasons.
Relevant to the content of this article, there are a few cycles that I want to mention that will crop up later in our discussion:
- The light-dark cycle
- The sleep-wake cycle
- The activity-rest cycle
- The feeding-fasting cycle
For now, keep that idea at the back of your mind as we open some other tabs.
The Wonderful World of Chronobiology
Rhythms exist everywhere in biology. While we typically have one sleep-wake cycle per day, our sleep occurs in several repetitive cycles of ~90 minutes, so there is a rhythmicity to these sleep stages. Another example of a repeating rhythm is the menstrual cycle, which of course has an approximate cycle length of ~28 days that repeats.
We have a scientific field that looks at phenomena that occur in specific rhythms: chronobiology. Think of chronobiology as the “biology of time and internal biological clocks.” Under the umbrella of chronobiology, we have different branches based on classifying rhythms according to their duration. In the above examples, we have:
- A rhythm lasting less than 24 hours (a sleep cycle), which we call an ultradian rhythm.
- A rhythm lasting more than 24 hours (the menstrual cycle), which we call an infradian rhythm.
So, as you have guessed by now, a rhythm of about 24 hours is what we define as a circadian rhythm. Just a few examples of biological processes that occur with a circadian rhythmicity include:
- Hormones – e.g. melatonin, cortisol, leptin, etc.
- Sleep-wake cycle – sleep anticipation in the brain default mode network (DMN)
- Core body temperature – reaches peak during the day and minimum point at night
- Immune system activity – e.g. pro-inflammatory cytokines like TNF
The implications of circadian biology for human health are difficult to understate. For the purposes of this discussion, we will only focus on a few elements.
Regulation of Circadian Rhythms
If we go up into the brain, and specifically into the hypothalamus, we have something called the suprachiasmatic nucleus (SCN). The SCN has ~ 20,000+ neuronal cells, which are responsible for circadian processes. The SCN can be thought of as our “master clock” or central circadian clock.
Biological (circadian) clocks produce circadian rhythms and regulate their timing. (Side bar: For the nerds of you in the audience who might be wondering, at a molecular level these clocks provide circadian timing through transcription/translation feedback loops involving the transcription factors CLOCK and BMAL1. If you want to get into specifics, this paper by Partch is a good start.)
One of the intrinsic characteristics of circadian clocks (biological clocks) is that they drive the circadian rhythm endogenously, meaning that the approximately 24-hour period previously mentioned will be maintained without any external influence (e.g. it will be maintained with conditions of constant darkness, for example, or across different temperatures). It is important to note that when I stated that circadian rhythms are “about 24 hours,” this is purposefully selected and precise language. Without the influence of external stimuli, these rhythms don’t run with an exact 24-hour period, but rather a bit longer or shorter than that. On average, in most humans, the endogenously generated period would be about 24 hours and 15 minutes, and so this leaves a slight mismatch between that rhythm period and our 24-hour day. Over time, the clock time of these peaks/troughs in circadian rhythms would be shifting more and more (we’ll revisit phase shifts later). And so, on the premise that we want alignment between these circadian rhythms and our 24-hour day, we need something to reset these rhythms to a more precise 24 hour period daily…
That “something” comes in the form of stimuli/signals from our environment. Certain stimuli have the ability to entrain (or “set”) circadian rhythms. In fact, we have a beautiful German word for such a stimulus: a zeitgeber (literally translated “time giver”). This process of a zeitgeber influencing the circadian rhythm is known as “entrainment.” Perhaps the most important zeitgeber is light. Specialized retinal cells in the eye contain a photo-pigment called melanopsin. This allows for the detection of light entering the eye (and as will be worth noting for later, melanopsin most effectively absorbs blue light). The SCN is actually attached to the retina, and so it can receive signals indicating when there is light present, allowing it to synchronize the body to the light cycle or solar day.
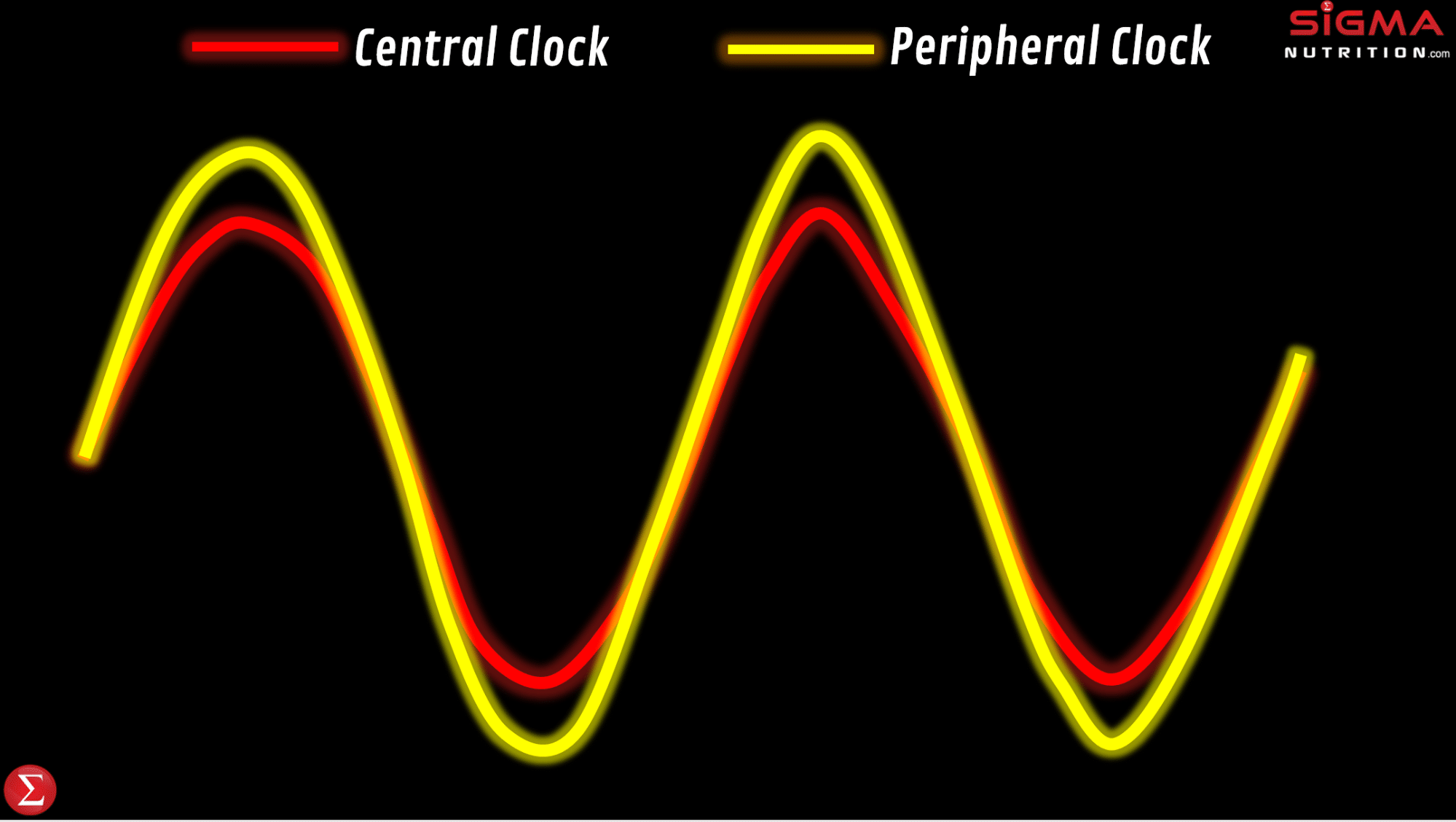
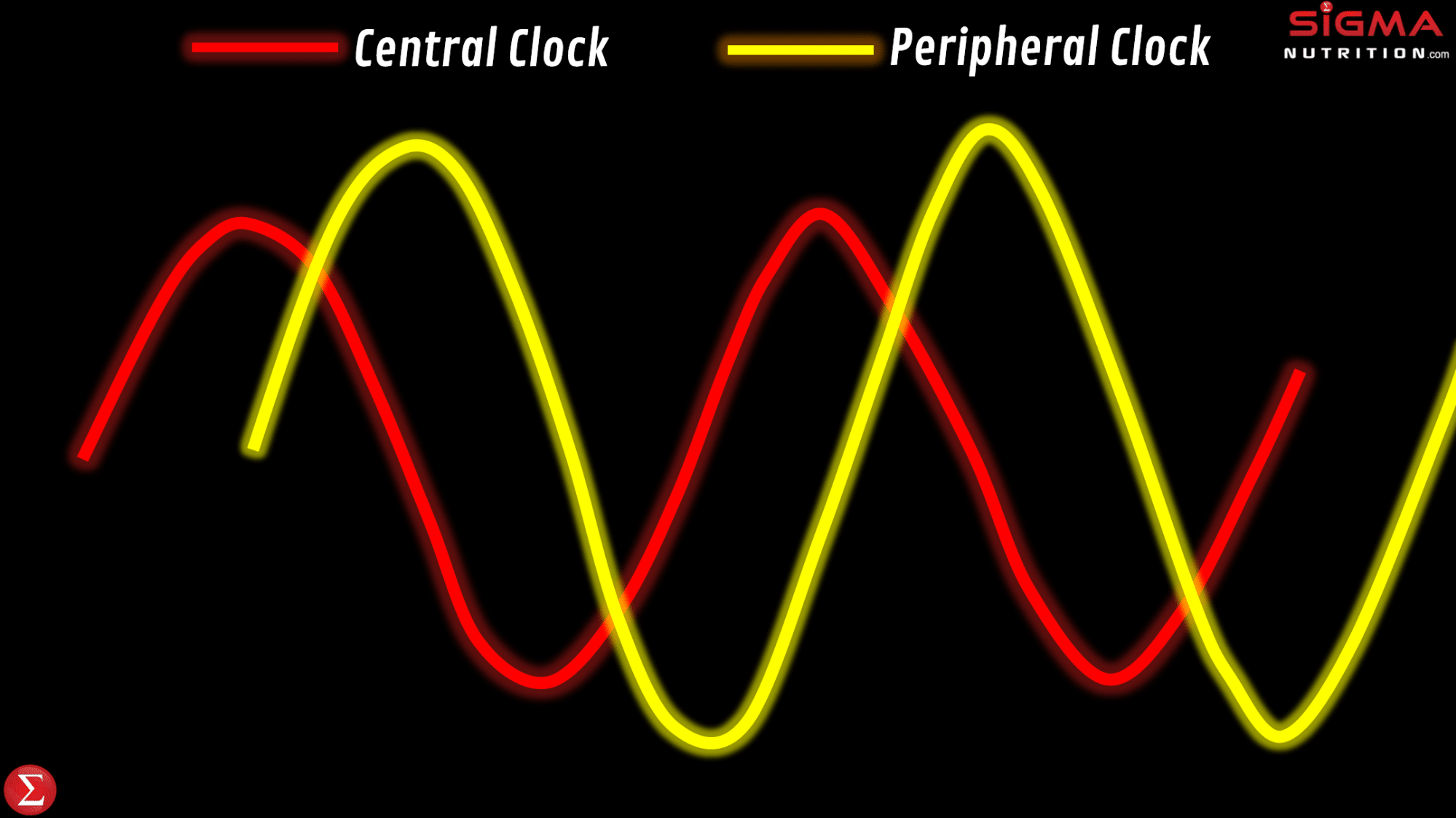
It’s common for people to colloquially reference their “body clock being off,” particularly in relation to jet lag or from having a sleep/wake time that is considerably different from their norm. Such comments are along the right lines. Time-zone travel and changes in sleep/wake times cause circadian phase shifts. However, we don’t just have one circadian clock; in addition to our master/central clock, there are circadian clocks located in tissues all around the body. These are referred to as peripheral clocks.
For example, we have peripheral circadian clocks in tissues of the gut, liver, pancreas, adipose tissue, and skeletal muscle. This opens up the potential for peripheral clocks to control the timing of digestion, nutrient metabolism, hormones, appetite and physical activity. Whilst peripheral clocks can be regulated by the master clock, studies done in tissue/cell culture models show rhythms persist in those cells even when detached from the SCN. This would indicate that these peripheral clock rhythms reflect local clocks and can be influenced by factors other than the SCN, meaning that peripheral clocks can be influenced by certain zeitgebers that don’t influence the master clock. This can independently influence the rhythmicity and timing of physiological processes in these specific tissues.
Circadian Phase Shifts & Chronodisruption
Adverse health effects can be driven by a misalignment between the master and peripheral clocks, relative to the external environment and behaviors. This desynchronization is defined as chronodisruption (or circadian disruption). “Chronodisruption” leads to serious health impairments in virtually every living organism in which it’s been examined. So to maintain health, we want to have a synchronization between our: a) master clock, b) peripheral clocks, and c) external environment that influences the biological clocks.
In chronobiology research, researchers make use of “phase shift protocols,” in which the light/dark cycle is either abruptly advanced or delayed. This temporarily desynchronizes an individual’s internal body clocks from the ambient light/dark cycle.
Unfortunately, many common aspects of modern life are misaligned with timing that would synchronize our clocks in an optimal fashion. For example, we know that light/dark cycles are the primary entrainers of the SCN and hence all circadian processes, but we have an environment where it’s possible to not only be exposed to bright light at night, but also to avoid sufficiently bright light in the early part of the day. Factor in work schedules, indoor environments (people in the US spend about 87% of their time in enclosed buildings), Daylight Savings, time-zone travel, etc., and we have a long list of societal factors that can cause chronodisruption.
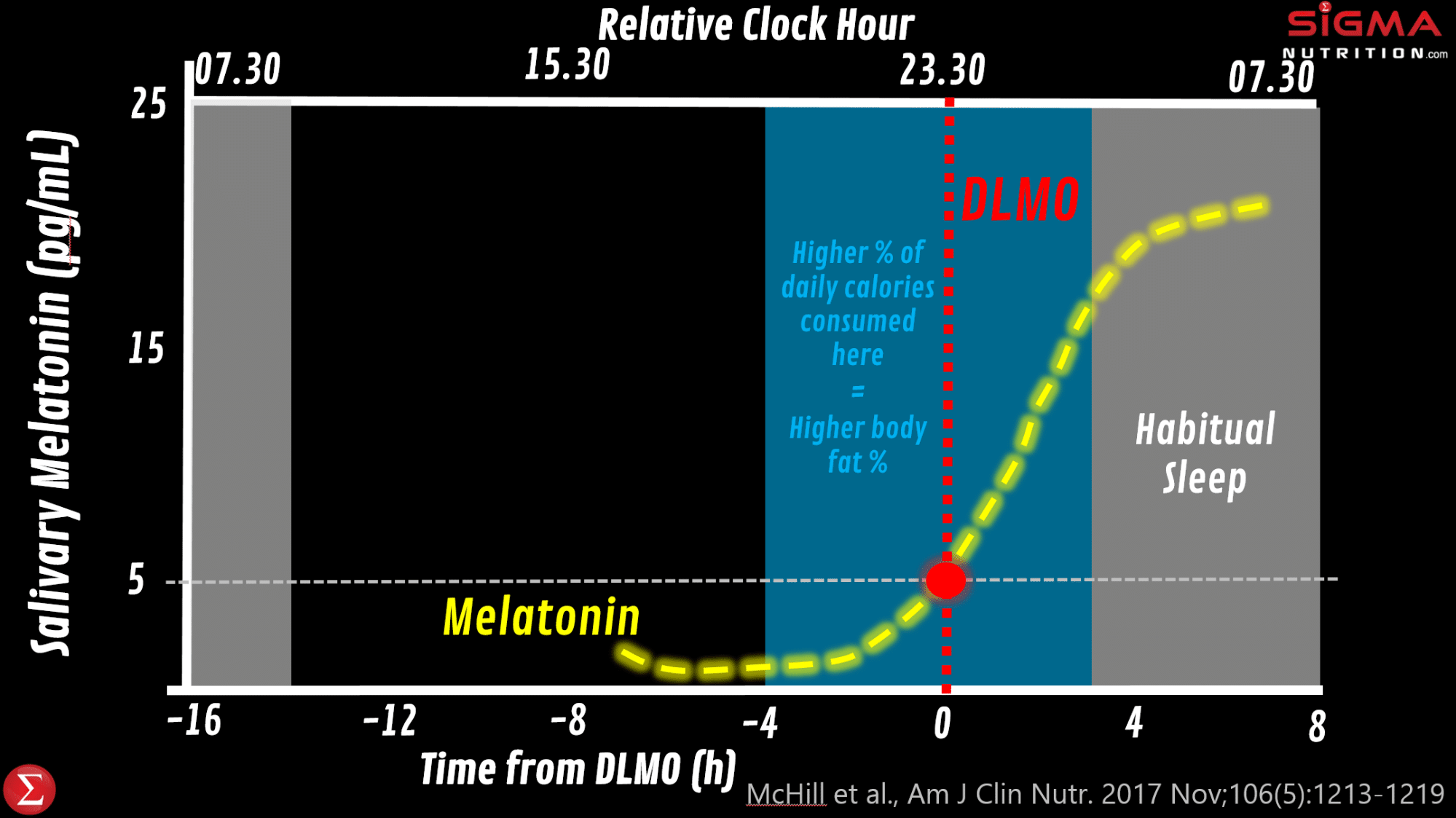
Light, in addition to fine-tuning the SCN, acts to inhibit melatonin synthesis. Because melatonin is metabolized rapidly, plasma melatonin levels are low during the day and high during the night. Dim light melatonin onset (DLMO) is the initial surge in melatonin release in the early part of the night under low light conditions. DLMO is a consistent and reliable measure of the intrinsic circadian phase. DLMO can therefore be used as a proxy for the onset of the biological night in humans, with melatonin offset in the morning corresponding to the start of the biological day. The timing of exposure to outdoor daylight plays a role in determining circadian phase and onset of sleep. Some of Till Roenneberg’s work has estimated that every additional hour spent outdoors may correspond to 30 minutes of phase advance.
If we consider the impact of circadian misalignment on our physiology, the best (albeit not the most eloquent) way to put it is: everything is screwed. Basically, if you’re sadistic and your goal is endocrinological destruction, then your tool of choice should be chronodisruption.
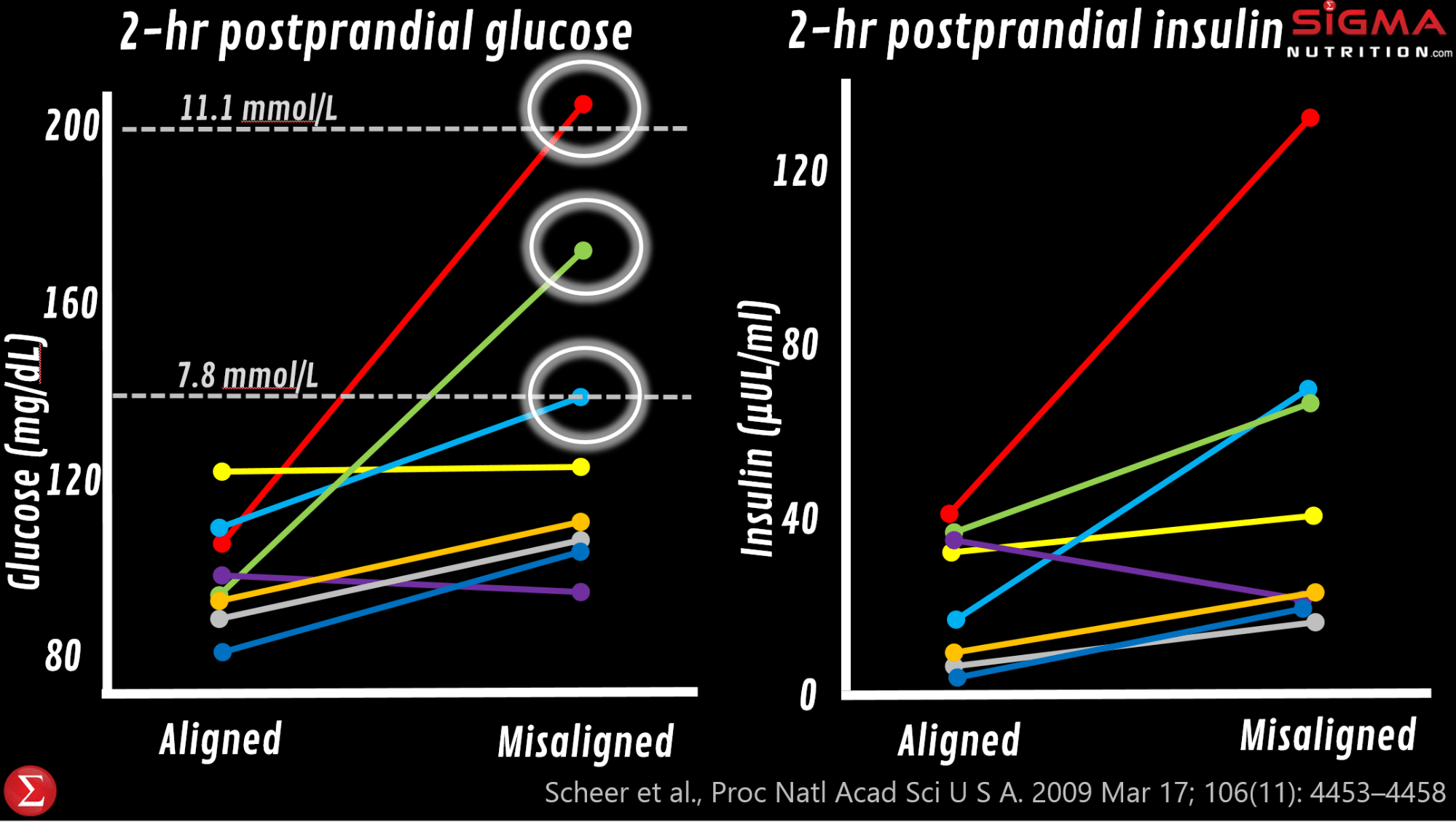
Excellent work from Frank Scheer’s lab shows that when someone is in circadian misalignment, they will have elevated glucose, elevated insulin, a completely flipped cortisol rhythm, significantly lower leptin levels (which tends to drive less activity and increased drive to eat), and reduced sleep efficiency.
In fact, in that study, 3 of the 8 subjects (figure below) saw their postprandial glucose response (which was in the normal/healthy range at baseline) end up measuring what would be indicative of a prediabetic or diabetic state (>7.8 mmol/L or >11 mmol/L) after becoming maximally misaligned (i.e. an 8pm, rather than 8am, wake-up time).
Chrononutrition: The Impact of Feeding
So what does this all have to do with our diet? As you might guess based on our discussion of chronobiology, chrononutrition refers to the interplay between nutrition and circadian biology. As I see it, there is a bi-directional relationship between diet and circadian biology:
- Impact of feeding on circadian rhythms: Nutrient ingestion/metabolism can entrain peripheral clocks.
- Impact of circadian rhythms on feeding (nutrient metabolism): Several biological processes related to metabolism, digestion, and hormone secretion have a circadian rhythmicity
Impact of feeding on circadian rhythms
One of the potential external factors that can entrain the peripheral clocks is nutrient ingestion. However, it seems that nutrient ingestion doesn’t have the same entraining effect on the SCN (master clock). Therefore, when and how we eat has the potential to synchronize or desynchronize our circadian rhythms, causing circadian alignment or misalignment. So perhaps timing feeding-fasting cycles in alignment with light-dark cycles can synchronize the biological and solar clocks.
Impact of circadian rhythms on feeding (nutrient metabolism)
Several biological processes related to metabolism, digestion, and hormone secretion have a circadian rhythmicity. Therefore, there may be “better” and “worse” ways to eat based on this diurnal variation. For example, we know that there is a diurnal pattern to:
- Gastric emptying – rates of gastric emptying peak in the morning.
- Beta-cell function – 15% higher in morning.
- Postprandial glucose response – Greater glucose excursions after eating occur in the evening vs. the same meal eaten in the morning.
- Diet-induced thermogenesis – Morris and colleagues found diet-induced thermogenesis to be 44% lower in evening vs. morning.
We also have evidence that chronodisruption alters appetite hormones and desire to eat.
Whilst the action of the master clock exerts a large influence on food intake, energy expenditure and insulin sensitivity, the peripheral clocks carry out additional controls. For example, the peripheral clocks located in gut tissue will regulate food absorption. Similarly, peripheral clocks in fat tissue regulate insulin sensitivity in the tissue, whilst peripheral clocks in the pancreas regulate the secretion of insulin.
So, if feeding can impact circadian clocks in the manner discussed so far, that leaves us with several interesting questions to explore:
- Does this lead to (pragmatically) significant impacts on:
- Energy balance?
- Nutrient metabolism?
- Metabolic health?
- If so, what is the magnitude of the effect?
- Therefore are there “best” times to eat? Are there times we should avoid?
- Does the length of the eating window matter? If so, what is the optimal length?
There are four sub-areas of chrononutrition that I feel are worth considering and that may be useful in leading us to some pragmatic conclusions:
- Meal timing.
- Energy (calorie) distribution.
- Eating pattern consistency.
- Feeding-fasting cycles.
Meal & Nutrient Timing
Are there better times to eat and/or are there times it’s best to avoid eating? A few lines of evidence shed some light on this. First of all, we see quite clearly and consistently that eating at biological night is a bad idea. Postprandial metabolism is much worse when a meal is eaten during the night (à la shift workers) than during the day. It’s likely that meals eaten close to DLMO are not the best idea from a metabolic standpoint. And of course, the larger the calorie (and potentially carbohydrate) load of the meal, the more detrimental it is to eat during the biological night.
We also reliably see tissues are more insulin sensitive in the morning than at night, with a decrease in insulin sensitivity across the day. Beta-cell function is also better in the morning (the relevance here being that insulin is secreted from the beta-cells of the pancreas, and thus beta-cell function is an important component of healthy glycemic control); therefore, there are greater glucose excursions following carbohydrate ingestion in the evening compared to earlier in the day. This may suggest that avoiding large carbohydrate loads in the evening or close to DLMO would be a good idea. There are several caveats here, which I’ll get to in the relevant section later in this article.
Observational work (image below) has shown that individuals consuming more of their daily calories between 4 hours before DLMO and the onset of sleep had a higher percentage of body fat.
So if we tentatively accept that eating meals (particularly big ones) at/close to biological night is not a great idea, what about the opposite end of the day? Is it important to eat something in the morning?
Theoretically, one could make a case that eating early in the day would be beneficial from the perspective of synchronizing our feeding with biological day or daylight hours. However, while I think that is fair reasoning, I’m uncertain about what this means for specific meal timing and if there is any need for a sense of immediacy around breakfast; i.e. how soon after waking would this first nutrient ingestion need to be? How large should the meal be? As of yet, there is no real way of answering these questions. And so let me use that lack of clarity to speculate on some potentially useful heuristics.
I do think there are likely benefits to eating early in the day. However, I am not convinced that should be synonymous with “eat as soon as you wake.” At the moment, I would probably think of it more like “eat something in the early part of the day,” which translates to probably several hours after waking (e.g. waking at 7:30am and waiting until 10:30am to have big breakfast seems perfectly fine to me, and is likely better than waiting until mid-afternoon). There are at least a few papers that hint at the idea that skipping eating until the afternoon might not be optimal from a metabolic or energy expenditure perspective. First, some research (this and this) suggests that skipping breakfast leads to worse glycemic excursions in later meals. However, there is the possibility that there is a “learned” effect at play; Thomas and colleagues found that the typical worsened glycemic excursion that occurs at lunch after skipping breakfast doesn’t occur in people who routinely skip breakfast everyday. This perhaps also suggests that it is consistency of timing, rather than the specific time, of that first meal that matters most (more on this later).
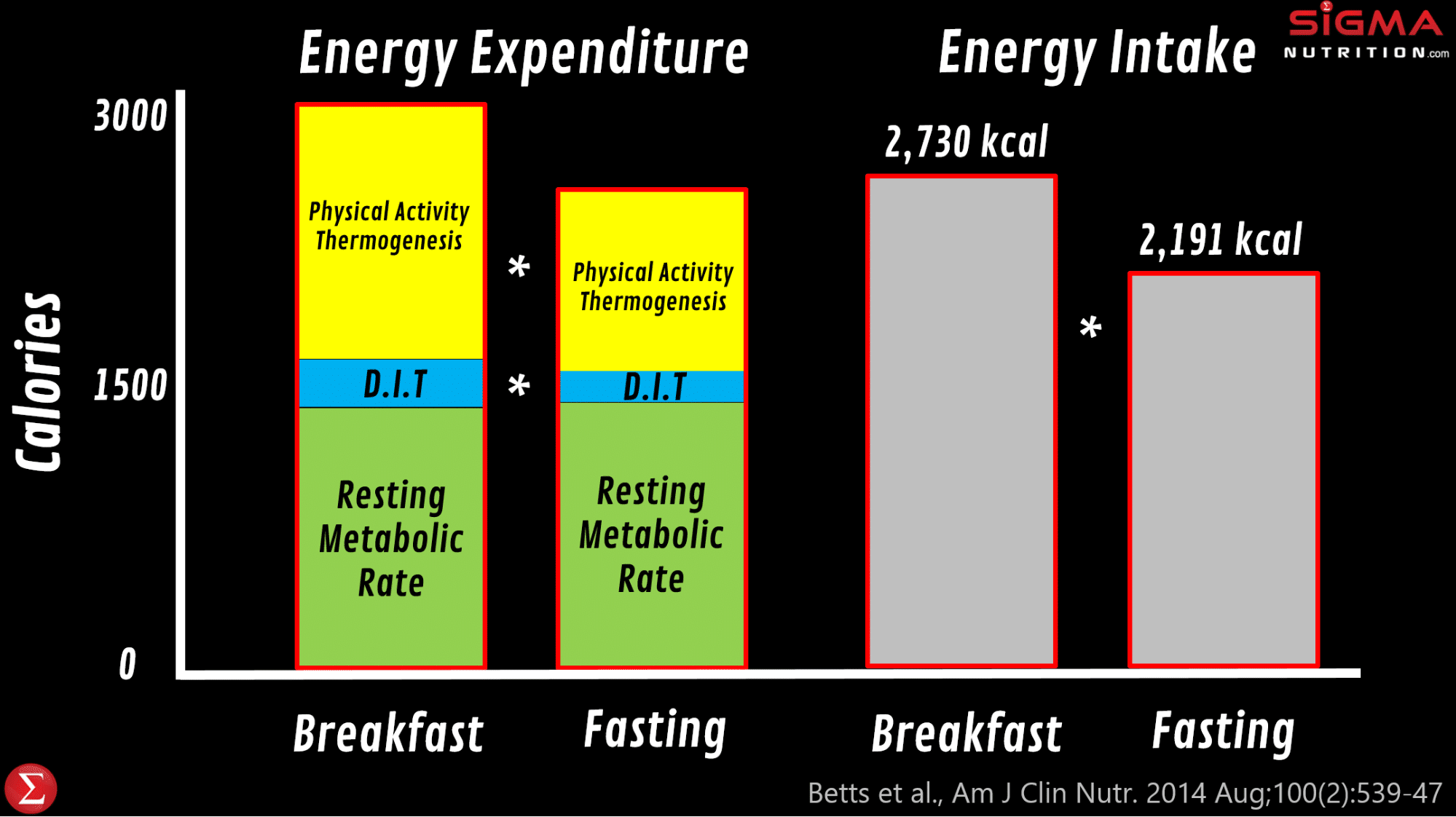
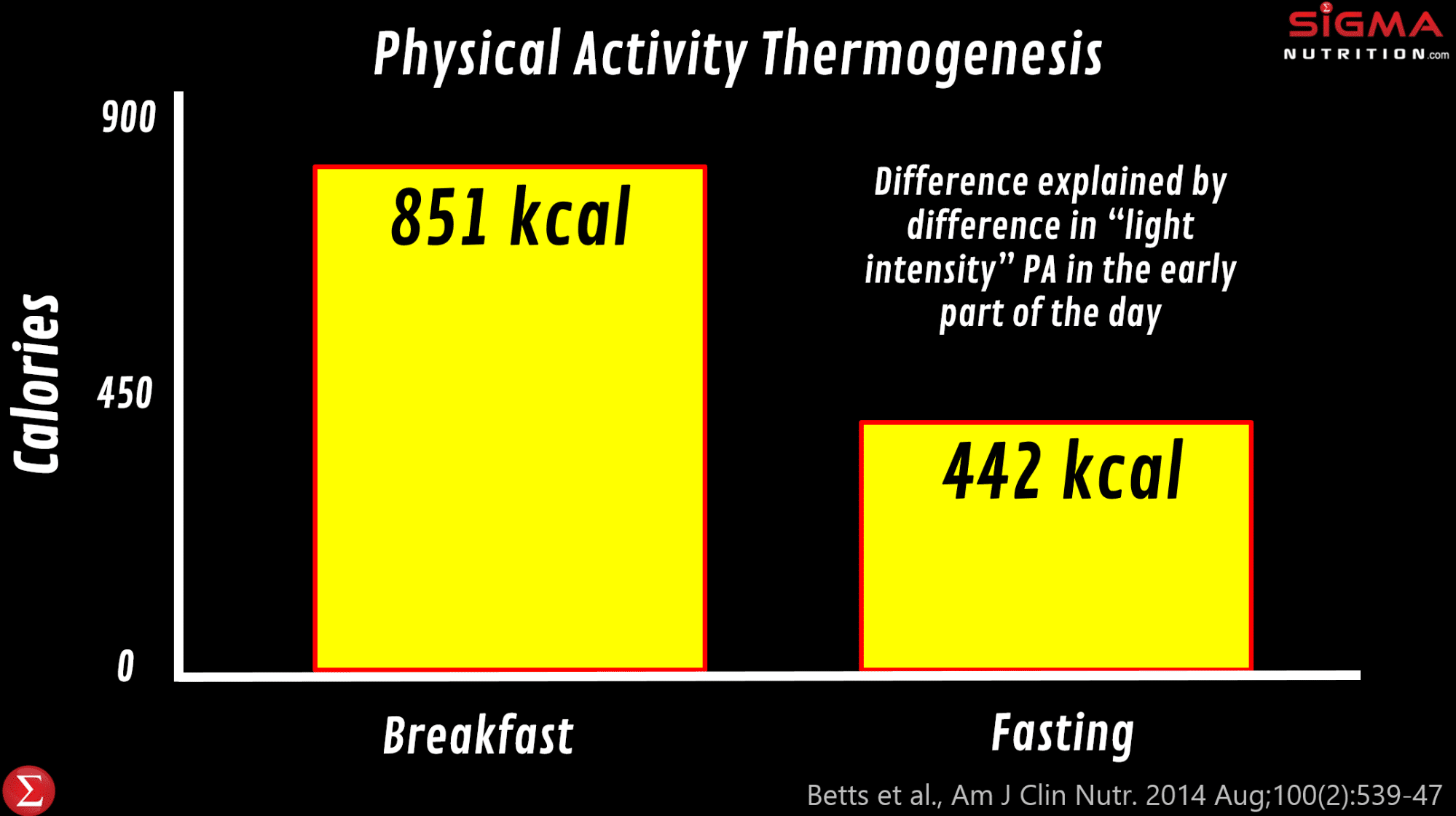
James Betts and colleagues at the University of Bath did a wonderfully insightful set of studies (the “Bath Breakfast Project”) that examined the impact of eating a large breakfast on energy balance. In one of their studies, they found that eating a relatively large meal (> 700 kcal) early in the day leads to much greater energy expenditure over the day compared to waiting until 12pm to have the first meal of the day. And most of this is explained by greater physical activity thermogenesis, specifically of light movement through the day (851 kcal vs. 442kcal ). And so whilst the breakfast group ingested more calories over the day than those fasting until 12pm (which is consistent with the intermittent fasting literature), there was no difference in energy balance and body composition; their increased calorie expenditure “cancelled out” the extra calories ingested. This suggests that it is at least plausible that we could see differences in energy balance (and therefore potentially body composition) via changing where meals are eaten. However, it should be noted that when the researchers repeated the study in obese participants (as opposed to lean participants), they found that physical activity thermogenesis was only different in the morning but not over 24 hours.
Energy (Calorie) Distribution
As per the McHill paper referenced earlier, it seems as though having a larger percentage of our daily calories late in the day (closer to sleep) could be a bad idea for body composition over time. Similarly, as I’ve already noted, it has been suggested that earlier in the day, we are physiologically better equipped to handle larger meals. Therefore, a reasonable hypothesis exists around the value of placing a higher proportion of our calories earlier in the day.
One of the most intriguing and often cited papers to suggest this to be of importance is a 2013 paper by Daniela Jakubowicz. In this study, the researchers compared the impacts of two diets, matched for calories and macronutrients, but that differed in the distribution of that intake. Both groups dieted on 1,400 kcal per day, eating 3 meals per day, with each meal (breakfast, lunch and dinner) being eaten at about the same time (within a 3-hour window) for all individuals. As for the distribution, participants were either assigned to what I’ll refer to as a “front-heavy” or “back-heavy” distribution. The front-heavy group had a large breakfast and small dinner, and the back-heavy group consumed a small breakfast and large dinner. Specifically:
- Front-heavy: 700 kcal breakfast, 500 kcal lunch, 200 kcal dinner
- Back-heavy: 200 kcal breakfast, 500 kcal lunch, 700 kcal dinner
In these calorie/macro matched conditions, a front-heavy distribution of a large breakfast and a small dinner led to significantly (both statistically & pragmatically) more weight loss (8.7 vs 3.6 kg) than a back-heavy distribution (small breakfast and large dinner). Now, I think it’s incredibly unlikely that such a large difference in body weight change between the groups could be explained solely through impacts of calorie distribution/timing on energy balance. There are almost certainly differences that could be assigned to accuracy of tracking, adherence, and behavior that weren’t captured in the study. This specific study didn’t provide participants with their meals (however, participants were provided with a specific meal plan). So it’s possible the reported intake wasn’t as accurate as we would hope. However, it does tell us that something is going on when the distribution of intake is changed, regardless of how much is physiological and how much is behavioral. Other notable differences included lower blood sugar excursions (lower glucose AUC) for the day as well as lower ghrelin (an appetite hormone) levels throughout the day for those front-loading their calories. So, there are differences in hunger, in blood glucose response, and body composition.
A pair of studies that came out of Marta Garaulet’s lab in Murcia, Spain also seem to support that a front-loaded distribution has benefits. First, a 2013 paper looked at the impact of when the largest meal of the day (which is lunch in a Mediterranean population like the one studied) is consumed. This was part of a 20-week weight loss intervention that included participants working with a dietitian, engaging in an exercise program, and also having behavioral change addressed. At the end of the study, they looked back at people’s logged food intake and split the participants into those that consumed their largest meal of the day before 3pm or after 3pm. Roughly half of the participants were before, and half were after. They saw that those who consumed their largest meal earlier (before 3pm) experienced greater weight loss across the trial and had less insulin resistance.
After seeing this difference, the same group did an intervention trial to actually assign people to an earlier or later calorie distribution. Again, it was focused on the time of the largest meal of the day (lunch). Over a two-week trial, they compared the effects of an early lunch (main meal eaten at 1:30pm) and a late lunch (main meal eaten at 4:30pm). They then gave the participants a test meal, testing their blood glucose response to the meal, and found that the early lunch resulted in a better postprandial glucose response.
Eating Pattern Consistency
It seems that the majority of people have “erratic” eating patterns with highly variable times their meals are consumed and frequencies of meals from day to day. Most people also eat later on the weekend, likely owing to later sleep and wake times on the weekend in those who work a typical work week. Gill and Panda (2015) refer to this change in eating time potentially causing “metabolic jetlag”; i.e., having a detrimental impact via shifting circadian clock phase. This is in parallel to the concept of “social jetlag” coined by chronobiologist Till Ronnenberg; social jetlag being the jetlag-like symptoms experienced by the chronodisruption of having very different sleep and wake time from the weekdays to the weekend (or any other set up, really). As sleep/wake times change (and thus timing of light exposure, activity, and eating), there are phase shifts in circadian clocks, therefore leading to the symptoms one experiences after traveling across time zones.
So, I feel there is a fair case to be made for the hypothesis that having relatively consistent meal times from day to day is likely beneficial. But what about meal frequency? One paper to look at this compared a fixed frequency of 3 main meals and 3 snacks per day vs. an irregular meal frequency with the number of meals alternating anywhere between 3 and 9 meals per day, every day for the 2 weeks of the study. The fixed meal frequency led to better postprandial glycemic responses after eating than the erratic nature of changing meal frequency everyday.
Feeding-Fasting Cycles (Time-Restricted Feeding)
One of the dietary interventions to sprout from chronobiology research is time-restricted feeding, whereby food intake is kept within a shortened window of time, therefore having a longer window of fasting. This setup is what most will think of when considering daily intermittent fasting protocols (for example, a daily 16:8 fasting:feeding regimen). What distinguishes time-restricted feeding as defined in research from other fasting regimens is its emergence from circadian biology.
Much of the early focus on TRF centered on animal studies. This data is overwhelmingly consistent in showing significant benefits in almost all contexts it has been studied in. For example, we see:
- A prolonged feeding period leads to fat gain, insulin resistance, and increased liver fat.
- When mice without a circadian clock are fed ad libitum, they gain weight. When feeding is kept within a 10-hour window during their active phase, they are protected against weight gain & metabolic syndrome.
- Giving access to a high-fat, high-sugar (obesogenic) diet when mice usually sleep tends to lead to greater body fat, compared to the same diet provided during the active phase (i.e. the “optimal” time from a circadian perspective). Doing so also flattens daily hormone rhythms and reduces energy expenditure and fat oxidation.
- If rodents are given access to a high-fat obesogenic diet but their feeding window is restricted to only 8-12 hours per day, even though it does not reduce overall caloric intake (compared to animals fed ad libitum), it still improves circadian rhythms and helps prevent or reverse metabolic diseases.
Beyond acknowledging that a LOT of good animal data exists and that overall it is very supportive of the premise that time-restricted eating is beneficial for metabolic health and body composition, it is probably more useful for us to place our focus on the human trials done to date. There are trials that vary between being more tightly and loosely controlled, with both being able to answer different questions. First let’s start with some studies high in external validity (i.e. give us an insight to pragmatic implications of using such strategies in the real world).
One study I like to bring up to start is a 10-week pilot trial that came out of the lab at the University of Surrey in the UK, because of how simple the diet intervention was for participants to understand and implement. In this trial, the only instruction participants were given was to a) delay their usual breakfast time by 1.5 hours, and b) bring forward their usual dinner time by 1.5 hours. There were no rules or guidelines about what foods to eat, how much to eat, etc. It was simply changing the times of the first and final meals. They found that people consumed significantly fewer calories and, in turn, lost weight. Of course, this trial couldn’t explain the mechanisms behind these findings, and there is always a high probability of people changing their normal meals and intake when enrolling in a nutrition study, regardless of being given instructions to or not. But pragmatically, this intervention was able to yield meaningful results (presuming weight loss is the goal), with no nutrition education, no ongoing interaction with a coach/dietitian, no specific recommendations about changing normal foods consumed and no recommendations to undereat.
In another intervention trial, participants were placed on a time-restricted eating regimen that consisted of an 8-hour feeding window. However, the 8-hour window could be self-selected, meaning each individual was free to place the window at whatever time of the day they wanted, as long as it remained at 8 hours. Similar to the Antoni study, there were no other food rules provided or recommendations about what foods or how much to consume. Over the course of the 16 weeks of the intervention, participants lost more than 3kg, and these changes in body weight were maintained at 1 year after follow-up.
In trials where time-restricted eating is compared to a control condition, we also see superiority of TRF for weight loss.
Based on what has previously been discussed in this article about the potential for timing of food ingestion to matter based on circadian biology, a natural question for circadian researchers to ponder is whether it matters what time of day the feeding window falls in? And if so, how much does it matter. Based on the mechanisms discussed early, one would hypothesize that a feeding window placed earlier in the day should lead to better outcomes.
In a 5-week crossover trial, researchers used an early time-restricted eating protocol (eTRF) where the feeding window was from 7am to 1pm. Compared to a control diet, the eTRF led to a lower postprandial insulin response, better beta-cell function, and less insulin resistance.
But what if we compare an eTRF protocol to a diet with the same feeding window duration, but placed later in the day? One study compared two different TRF protocols in a 7-day crossover trial. Both protocols had a 9-hour feeding window, but this was either placed from 8am to 5pm (early TRF) or from 12pm to 9pm (delayed TRF). In this trial, there were no major differences, although mean fasting glucose was slightly better in the eTRF condition. Both conditions led to significantly improved 3 hour PP glucose response compared to baseline.
Heuristics & Pragmatic Implementation
Collectively, all the research discussions to this point provides us with some guidance for practical application:
- When we eat a meal (relative to social clock time) has health implications.
- It may be beneficial to avoid eating at biological night.
- Biasing more calories to earlier, rather than later, in the day is superior for metabolic health and potentially body composition.
- A restricted eating window is beneficial for health and/or body composition.
- We should match the feeding window with biological day, wakefulness, and activity.
Heuristics that will likely result in benefits (for many):
- Avoiding eating during biological night.
- Have a restricted feeding window (maybe start with <12 hours per day. Most human data examines an 8-hour feeding window, but no ideal is yet known).
- Get daylight exposure early in the biological day. Avoid artificial light at night.
- Bias towards a “front-heavy” calorie distribution (i.e. don’t eat a high proportion of you daily calories in the late evening).
- Avoid meals, particularly those high in fat and/or carbohydrates, close to DLMO (or say at least ~2-3 hours pre-sleep).
- Avoid erratic eating: have consistent meal times and meal frequency from day-to-day.
Caveats, Exceptions & Contraindications
Now I’m sure after reading the list of proposed heuristics, many of you are thinking of several cases where those recommendations would not only be ineffective, but flat out counter-productive. And you’re correct. As heuristics, they of course cannot account for the many caveats and exceptions that may exist. There are four primary cases where some of these heuristics may be contraindicated:
- Athletes
- Resistance training in the evening
- Social occasions & lifestyle
- Ease of implementation and adherence
Athletes
In order to maximize athletic performance, there are several nutrition strategies that may be different from those applied to maximize human health. Many heuristics we commonly apply to the average person for health reasons may either be irrelevant, or even detrimental, to an athlete focused on performance. For example, frequently drinking sugar-sweetened beverages is something we would advise health-conscious individuals against. For certain athletes, there are specific times where we would actively encourage the consumption of a sugar-laden sports drink. With respect to the heuristics suggested in the previous section, there are several cases where they may not apply or be counter to evidence-based sports nutrition practices. Just some hypothetical examples include:
- If a bodybuilder’s priority is to maximize the amount of muscle mass they build in the next block of time, is a restricted feeding window their best tool? Or would they (at least theoretically) perhaps benefit from having an extra protein-feeding opportunity in the day?
- If an MMA athlete trains twice per day at high intensity, after their evening training session, is it likely conducive to their performance and recovery to tell them “let’s avoid eating (especially carbohydrates) at night”?
- If an athlete is struggling to consume enough calories (e.g. athlete attempting to gain weight, or an elite swimmer needing 8,000 kcal/d), is it wise to use a time-restricted eating model? Or avoid large meals in the evening?
Resistance training in the evening
This is a potential caveat that I likely suspect influences readers of this site. With muscle contraction from a sufficient stimulus (let’s say via lifting some weights), a glucose transporter (GLUT4) usually embedded in a muscle cells, moves to the surface of the cell. This transporter now makes it possible for glucose to move from the bloodstream into the muscle cell and, importantly, this process it NOT reliant on the action of insulin, as glucose disposal usually is (to be clear, insulin does stimulate GLUT4 translocation, but contraction-mediated GLUT4 translocation doesn’t require insulin). So the result is that we can now get glucose disposal from the bloodstream to the muscle cell (and hence bring down blood glucose levels) without needing insulin to mediate this process. This is relevant because earlier in this article, I discussed why we could hypothesize that consuming a carbohydrate-based meal would be more problematic late in the day compared to the morning, based on the mechanistic rationale that insulin sensitivity declines across the day and that beta-cell function is higher in the morning. But one interesting question to ponder is: how much would placing a resistance training session in the evening mitigate the potential problem? Because now, we can consume carbohydrates in the evening and get adequate glucose disposal, regardless of the cell’s insulin sensitivity status.
That said, there are other issues to consider with large meals late in the day (e.g. impacts on peripheral circadian clocks, impacts on sleep, impact of energy expenditure across the day). But at least there is a reason to suggest that we can mitigate the glycemic concerns of later carbohydrate feedings through the inclusion of evening training sessions.
Social occasions and lifestyle
This one is more of a pragmatic, behavioral, and emotional caveat. Essentially, while we could make a case that an early TRF protocol, with a bias toward more calories earlier in the day, would be “best” from a metabolic and physiological perspective, I can think of few things worse than missing out on a meal out with a spouse, drinks with friends, a family party, or any other valuable social occasion because you’re deathly afraid of any disturbance to you body’s circadian clocks. So yes, I believe there is value to, in general, thinking about circadian biology and how your usual eating may influence it, but also realize there are tremendous health benefits to being socially connected, enjoying oneself, and having happy memories with people you care about. It’s a similar issue with sleep. Most of the time, you should absolutely prioritize having behaviors that are conducive to getting plenty of sleep and maximizing the quality of that sleep. But don’t let the knowledge you have about the dangers of sleep restriction keep you from having the occasional late night if it’s in the name of a social occasion that is important to you and that will bring you happiness.
It’s also important to acknowledge that an individual’s usual lifestyle and schedule will likely impact which heuristics they can employ and/or to what extent. The obvious example is someone doing shift work (which, while a related issue to that covered in this article, is something that deserves its own dedicated article). Or consider a parent who has a family that only gets to eat together at a 7pm dinner when everyone is home together; do we really need to shove the Sutton paper into their face and say “you should try having your final meal at 1pm” in the name of #science? (Side note: That extreme example is actually the antithesis of science or evidence-based thinking, but I will remain off my soapbox on that until another day).
Adherence or ease of implementation
A commonly voiced concern with some of these heuristics is that they will make things unnecessarily difficult for people. And if it’s more difficult to time meals differently, then they won’t adhere to it. And if they won’t adhere to it, then what’s the point? All that thinking is logical and reasonable.
My default position has been: if trying to implement some of these “circadian strategies” undermines an individual’s ability to adhere to the fundamental pieces of healthy nutrition (e.g. if it worsens food quality, if it makes it more difficult to eat an appropriate amount of calories, if it causes stress, etc.) then of course, it’s unlikely something I’d recommend persisting with.
However, with that said, I suspect that the difficulty of making some of these changes is either not as high as what many assume or it is short-lived. Most of the TRF interventions seem to suggest it’s a very easy intervention for people to implement. And given how easy it is to understand, and the lack of attachment to food rules, I think it seems reasonable to suggest it may have potential to be scalable at a population-wide level.
One of the common objections from an adherence perspective is from those who enjoy eating larger meals late in the day. Oftentimes, such individuals enjoy a daily intermittent fasting protocol, skipping eating until the early afternoon and in many cases “saving” as many calories as they can for their final meal(s) late in the day. This is certainly appealing to many. And I identified as one of these fans of big, late meals, so I totally get it. But after changing around some of my diet habits to ones where I no longer have a large proportion of calories late in the day, I’ve found those preferences I thought I had were just learned behavioral ones, rather than preferences innate to me. Of course, I’m not suggesting this will be the case for everyone. But rather, I’m saying that if you remain open-minded, you may learn to be just as content with a different calorie distribution and meal timing if you give it a short trial.
Concluding Remarks & Next Steps
“We need more high-quality human trials!” I hear you cry! And yes, I get you. Correct, we need more high-quality human trials. Namely, to investigate things like:
- Circadian rhythms in different components of EE (RMR vs. NEAT vs. TEF).
- What magnitude of effect is attributable to actual circadian impacts on metabolism/energy balance and what is attributable to behavioral changes caused by intervention/circadian phase shifts.
- What is the exact mechanism by which greater weight loss is occurring with earlier versus later eating?
However, I would caution against allowing this uncertainty to lead us to conclude we don’t know enough to start implementing chrononutrition principles into practice. Scientific thinking is thinking in probabilities, not certainties. We have remaining questions, and they are important questions to get answers to. So for right now, when making a decision for how important this is in practice, I see this as evaluating all the lines of evidence we’ve looked in this article, and using the weight of each type of evidence to conclude the probability of something being likely correct. What picture is emerging? We know circadian rhythm disruption is bad for health. Mechanistic research shows feeding can entrain peripheral clocks. Animal data shows clear impacts on body comp, chronic disease risk, etc. Observational data in humans links “bad” feeding schedules with poorer outcomes (e.g. eating late at night, shift work).
One of the hallmarks of a relatively new field of research (or perhaps all fields of research!) is that for every interesting observation made, it leaves us with several-fold more questions. At this stage, the evidence base in the field of chrononutrition has shown us that circadian biology should be a consideration in nutrition decisions, and it has given us several very interesting observations and hypotheses. But, as of right now, several important questions remain, including some you may have right now. For example, how does someone’s chronotype influence the “correct” timing? (A study from earlier this year suggests a “chronotype-adjusted diet” is more effective than the traditional hypocaloric dietary treatment in reducing bodyweight, BMI, and waist circumference [Galindo Munoz et al., 2019]. There are some issues with this paper, but we don’t have time for that now).
Here’s just a small list of some big questions:
- What is the optimal length of a feeding (or fasting) window?
- When should we eat the first meal and how much does it matter?
- How important is calorie distribution across the day?
- What is the impact of calorie distribution and meal timing on energy expenditure?
- What aspects of energy expenditure are responsible for differences?
- How is calorie intake impacted? By what mechanisms?
TL;DR
- Circadian biology plays a fundamental role in human health.
- Research has shown that nutrient ingestion can impact our “body clocks” in peripheral tissues around the body, suggesting that when we eat our meals can have implications for health via influencing circadian rhythms.
- In addition, it has been hypothesized that having a restricted feeding window (time-restricted feeding) can have beneficial impacts on body composition and health, likely via circadian effects at least in some part.
- A related hypothesis suggests that the distribution of calories over the day (majority eaten early vs. late) can also have health impacts.
- My personal interpretation of the current literature available leads me to tentatively conclude that, in general, the following heuristics would be beneficial for many people to follow: 1) avoid eating during biological night, 2) avoid meals, particularly those high in fat and/or carbohydrates, close to DLMO (or say, at least ~2-3 hours pre-sleep), 3) bias more calories to earlier in the day (i.e. don’t eat a high proportion of your daily calories in the late evening), 4) have consistent meal times and meal frequency from day-to-day, 5) have some restricted feeding window (start with <12 hours per day, but no ideal is yet known), 6) get daylight exposure early in the day and avoid artificial light (blue and green wavelengths of light specifically) as much as is pragmatically reasonable at night.
- There are several caveats and exceptions to the above heuristics. Implications may be different for athletes, for those trying to gain weight, for those who such heuristics undermine adherence to nutrition fundamentals, and in situations where social interactions and fun should be prioritized.
Bonus Material
You can watch a lecture I gave on the topic of chrononutrition here.